Reference: March 2026 | Issue 3 | Vol 12 | Page 34
Heart failure with preserved ejection fraction (HFpEF) has emerged as one of the most significant clinical challenges in contemporary cardiovascular medicine. Once considered a benign condition relative to heart failure with reduced ejection fraction (HFrEF), HFpEF now accounts for more than half of all heart failure cases worldwide, with its proportion continuing to rise year on year.
Epidemiological data demonstrated a clear temporal increase in the proportion of heart failure patients classified as HFpEF, reflecting an ageing population burdened by hypertension, obesity, diabetes mellitus, and sedentary lifestyles.1
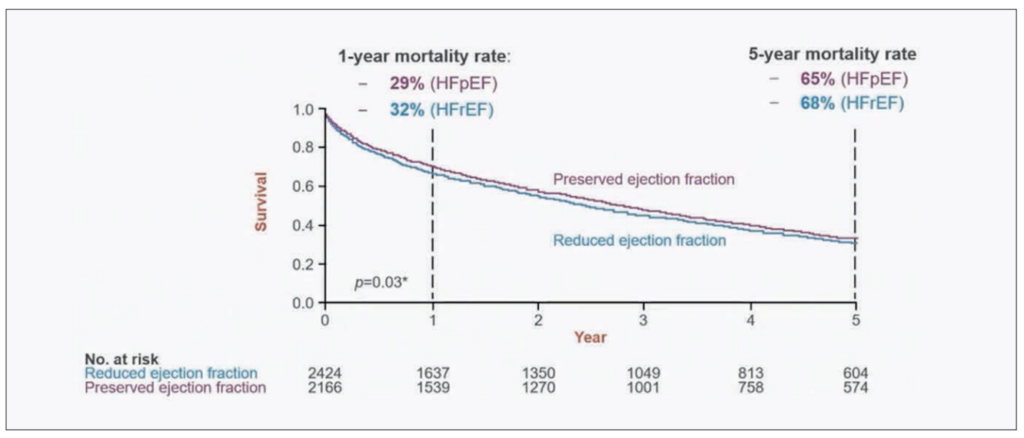
Perhaps most importantly, and contrary to earlier assumptions, mortality in HFpEF is comparable to that observed in HFrEF. Landmark studies revealed similar mortality trends across the ejection fraction spectrum, firmly establishing HFpEF as a condition demanding equal clinical attention and therapeutic innovation (Figure 1).2
Despite this, for decades, clinicians had no evidence-based pharmacological therapy that convincingly improved outcomes in HFpEF. That landscape has changed dramatically in recent years, and this article provides a comprehensive overview of the current state of diagnosis, management, and the rapidly evolving trial evidence base for HFpEF.
Classification of heart failure by ejection fraction
The 2021 European Society of Cardiology (ESC) guidelines formally categorise heart failure into three groups based on left ventricular ejection fraction (LVEF) (Figure 2).3 Heart failure with reduced ejection fraction (HFrEF) is defined by an LVEF of 40 per cent or below, with the presence of symptoms and/or signs.
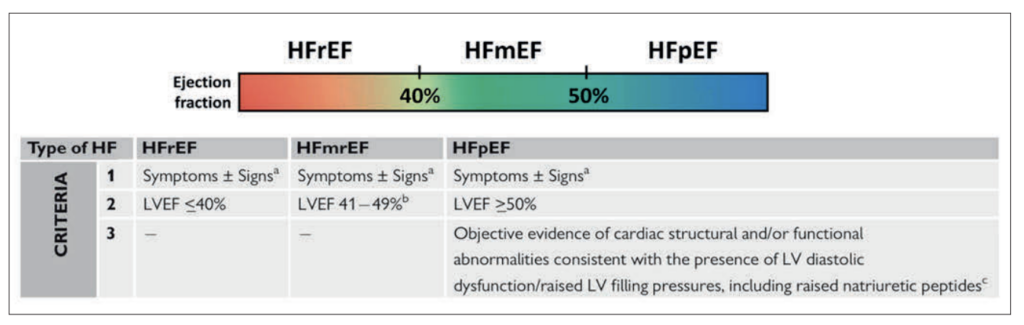
Heart failure with mildly reduced ejection fraction (HFmrEF) encompasses patients with an LVEF of 41 to 49 per cent. HFpEF is defined by an LVEF of 50 per cent or above, together with symptoms and/or signs and objective evidence of cardiac structural and/or functional abnormalities consistent with left ventricular diastolic dysfunction or raised filling pressures, including elevated natriuretic peptides.
This tripartite classification is clinically important because each phenotype carries distinct pathophysiological underpinnings, prognostic implications, and, increasingly, distinct treatment strategies.
Pathophysiology of HFpEF
Unlike HFrEF, where the heart muscle weakens and dilates, in HFpEF the heart contracts normally but struggles to relax and fill properly. A normal ejection fraction can therefore be misleading – patients often have a thickened, stiff ventricle, an enlarged left atrium, and elevated filling pressures that cause breathlessness on exertion.
The driving force behind this is now understood to be chronic systemic inflammation fuelled by common comorbidities such as obesity, diabetes, hypertension, and chronic kidney disease. This inflammation damages the coronary microvasculature, stiffens the heart muscle, and promotes fibrosis – a concept known as the microvascular inflammatory hypothesis – and it explains why therapies targeting inflammation, metabolism, and obesity are now showing such promise in HFpEF.4
Current diagnostic algorithms
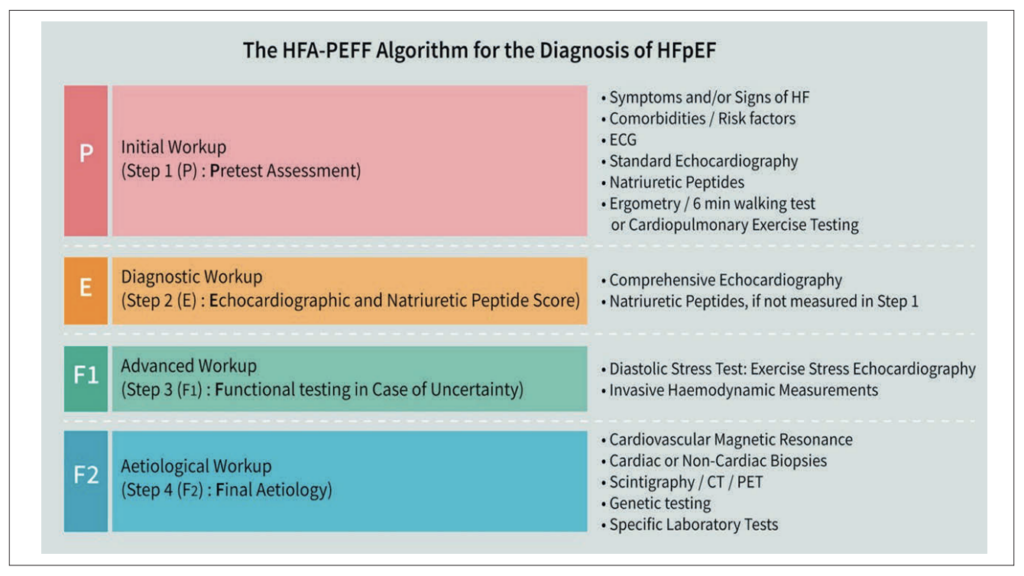
Diagnosing HFpEF in clinical practice remains challenging, as many patients present with non-specific symptoms such as exertional dyspnoea, fatigue, and exercise intolerance in the setting of a preserved LVEF. Two validated diagnostic scoring systems have been developed to assist clinicians – the H2FPEF score and the HFA-PEFF algorithm.
The H2FPEF score
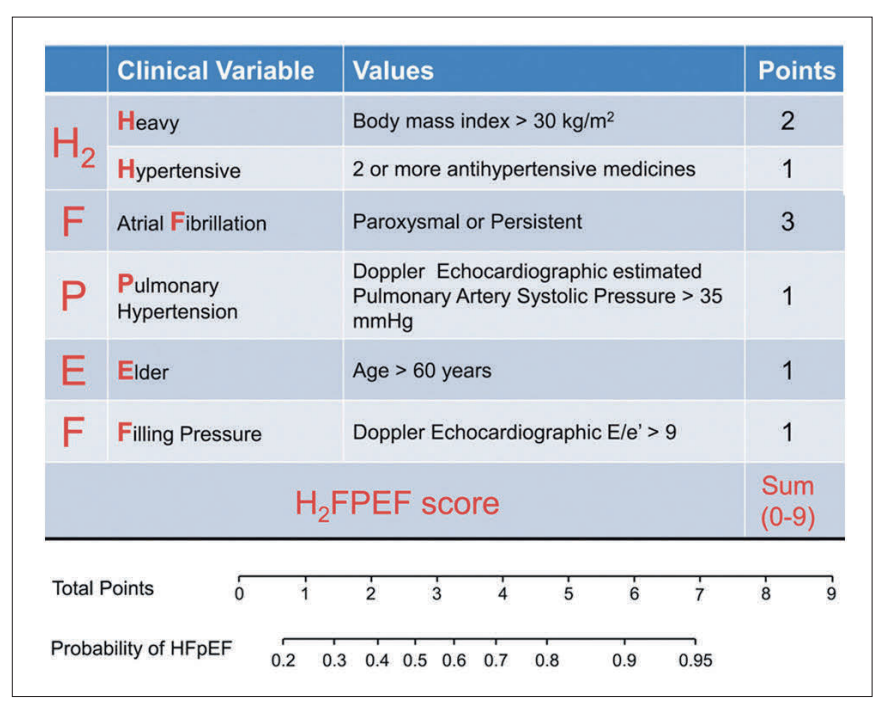
The H2FPEF score is a clinical prediction tool derived from invasive haemodynamic data (Figure 3).5 It incorporates six variables – body mass index (heavy), use of two or more antihypertensives (2 hypertensive), atrial fibrillation (AF), pulmonary artery systolic pressure (pulmonary hypertension), and E/e’ ratio (filling pressure) – generating a composite score from zero to nine. A score of six or above is highly suggestive of HFpEF, while a score of two or below makes the diagnosis unlikely.
The HFA-PEFF algorithm
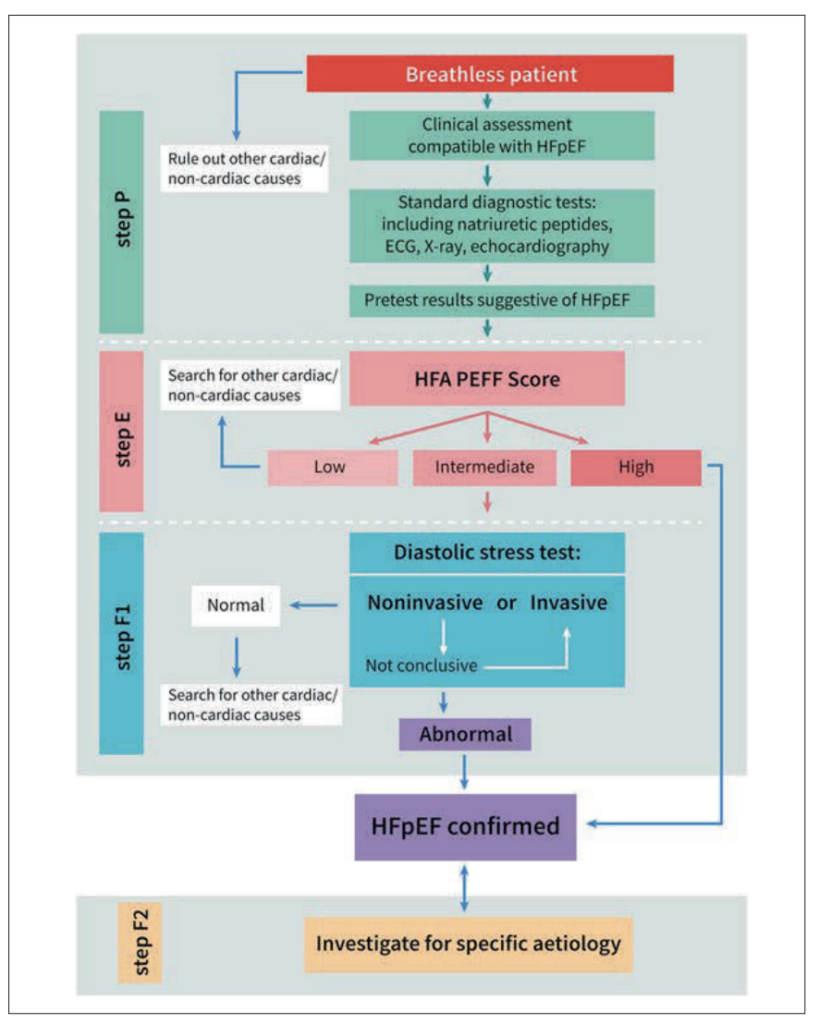
The HFA-PEFF algorithm, developed by the Heart Failure Association of the ESC, provides a structured stepwise approach to diagnosing HFpEF (Figure 4).6 Beginning with a clinical pre-test assessment (Step P), it progresses to echocardiographic and natriuretic peptide scoring (Step E), where major criteria score two points and minor criteria one point across functional, morphological, and biomarker domains (Figure 5).
A score of five or above confirms HFpEF; one or below makes it unlikely; and intermediate scores (two-four) require diastolic stress testing (Step F1). Once confirmed, Step F2 investigates for specific aetiologies such as cardiac amyloidosis or hypertrophic cardiomyopathy.
Evolution of treatment: From frustration to evidence
The treatment landscape for HFpEF has undergone a remarkable transformation over the past two decades. A review of successive ESC heart failure guidelines illustrates this evolution vividly.
ESC Guidelines: A 20-year journey
The 2001 ESC heart failure guidelines made virtually no specific therapeutic recommendations for patients with preserved ejection fraction.7 By 2012, guidelines acknowledged HFpEF as a distinct clinical entity, but conceded that no treatment had been convincingly shown to reduce morbidity or mortality, with management limited to treating underlying comorbidities.8 The 2021 guidelines advanced further, recommending diuretics for congestion, weight reduction in obese patients, and exercise training to improve symptoms, but disease-modifying pharmacological options remained absent.3
The pivotal shift came with the 2023 ESC Focused Update, which incorporated the results of landmark sodium-glucose co-transporter-2 inhibitor (SGLT2i) trials and provided, for the first time, a Class I recommendation for a specific pharmacological therapy in HFpEF.9 This update marked a watershed moment in the field and has been followed by further practice-changing trial results.
SGLT2i: The first pillar of HFpEF therapy
EMPEROR-Preserved
The EMPEROR-Preserved trial was the first large-scale randomised controlled trial to demonstrate a significant reduction in the primary composite endpoint of cardiovascular death or heart failure hospitalisation in patients with HFmrEF and HFpEF.10
The trial enrolled 5,988 patients with chronic heart failure, NYHA class II-IV, and an LVEF above 40 per cent. Empagliflozin 10mg daily reduced the primary endpoint with a hazard ratio of 0.79 (95% CI 0.69–0.90, p<0.001). The benefit was driven predominantly by a reduction in heart failure hospitalisations, with no significant effect on cardiovascular mortality as an individual endpoint.
DELIVER
The DELIVER trial evaluated dapagliflozin 10mg daily in 6,263 patients with symptomatic heart failure and an LVEF above 40 per cent.11 The primary composite endpoint of worsening heart failure or cardiovascular death was significantly reduced, with a hazard ratio of 0.82 (95% CI 0.73–0.92, p<0.001).
DELIVER strengthened the evidence base by demonstrating consistent benefit across the HFmrEF and HFpEF spectrum, irrespective of diabetes status, and established SGLT2i as a foundational therapy across the heart failure continuum.
On the basis of these two pivotal trials, the 2023 ESC Focused Update recommended SGLT2i (empagliflozin or dapagliflozin) with a Class I, Level A recommendation for patients with HFmrEF, and a Class I recommendation for symptomatic HFpEF patients.9 This represented the first Class I pharmacological recommendation in HFpEF and has transformed clinical practice globally.
MRAs: Steroidal versus non-steroidal
Mineralocorticoid receptor antagonists (MRAs) have long been a cornerstone of HFrEF management. However, the steroidal MRAs (spironolactone and eplerenone) have shown inconsistent results in HFpEF. The TOPCAT trial, which evaluated spironolactone in HFpEF, was complicated by significant regional heterogeneity in patient recruitment, and while post-hoc analyses of the Americas cohort suggested benefit, the overall trial result was neutral for the primary endpoint.12
The advent of non-steroidal MRAs represents a major pharmacological advance. Finerenone, a non-steroidal MRA, differs from traditional agents in several important ways. It exhibits higher mineralocorticoid receptor selectivity and potency than spironolactone and eplerenone.
As an inverse agonist rather than a partial agonist, finerenone demonstrates more complete receptor blockade. Its tissue distribution is balanced between the heart and kidneys (in contrast to the renal-predominant distribution of steroidal MRAs), it has no central nervous system penetration or sexual side effects, and it exerts a modest effect on blood pressure and a lower risk of hyperkalaemia.
The FINEARTS-HF trial
The FINEARTS-HF trial was a pivotal randomised, double-blind, placebo-controlled trial evaluating finerenone in 6,016 patients with HFmrEF or HFpEF (NYHA class II–IV, LVEF ≥40%). Over a median follow-up of 32 months, finerenone significantly reduced the primary composite endpoint of cardiovascular death or total heart failure events, with a rate ratio of 0.84 (95% CI 0.74–0.95, p=0.007).13
This translated to an absolute rate reduction of 2.8 events per 100 patient-years. The FINEARTS-HF results represent the second major pharmacological breakthrough in HFpEF, establishing finerenone as a potent addition to the therapeutic armamentarium.
The clinical impact of FINEARTS-HF has been swift. The JCS/JHFS (Japanese Circulation Society/Japanese Heart Failure Society) 2025 guidelines have already incorporated finerenone with a Class IIa, Level B-R recommendation for HFpEF, stating that in symptomatic HFpEF patients, an MRA (finerenone) can be considered to reduce the risk of cardiovascular death or heart failure exacerbation events.14
Steroidal MRAs (spironolactone, eplerenone) received a Class IIb recommendation in the same guidelines. A European guideline update incorporating FINEARTS-HF is anticipated in the near future.
Angiotensin receptor-neprilysin inhibition: PARAGON-HF
The PARAGON-HF trial evaluated sacubitril-valsartan versus valsartan in 4,822 patients with HFpEF (NYHA class II–IV, LVEF ≥45%). While the primary composite endpoint of heart failure hospitalisation and cardiovascular death narrowly missed statistical significance, subgroup analyses suggested benefit in women and in patients with an LVEF closer to the lower boundary (45-57%). Based on this evidence, current guidelines assign sacubitril-valsartan a Class IIb recommendation in HFpEF patients with LVEF below normal, positioning it as a potential option in selected patients.
GLP-1 RAs: A new frontier in obesity-related HFpEF
The emergence of glucagon-like peptide-1 receptor agonists (GLP-1 RAs) as a potential treatment for HFpEF represents one of the most exciting developments in the field. GLP-1 is an incretin hormone with pleiotropic effects extending far beyond glycaemic control. In the cardiovascular system, GLP-1 RAs promote cardioprotection, increase cardiac output, enhance vasodilation, reduce fatty acid metabolism, and decrease systemic inflammation.
Additional effects include weight loss through appetite suppression and delayed gastric emptying, improved renal function through natriuresis and diuresis, and reduced hepatic steatosis.
STEP-HFpEF and STEP-HFpEF DM
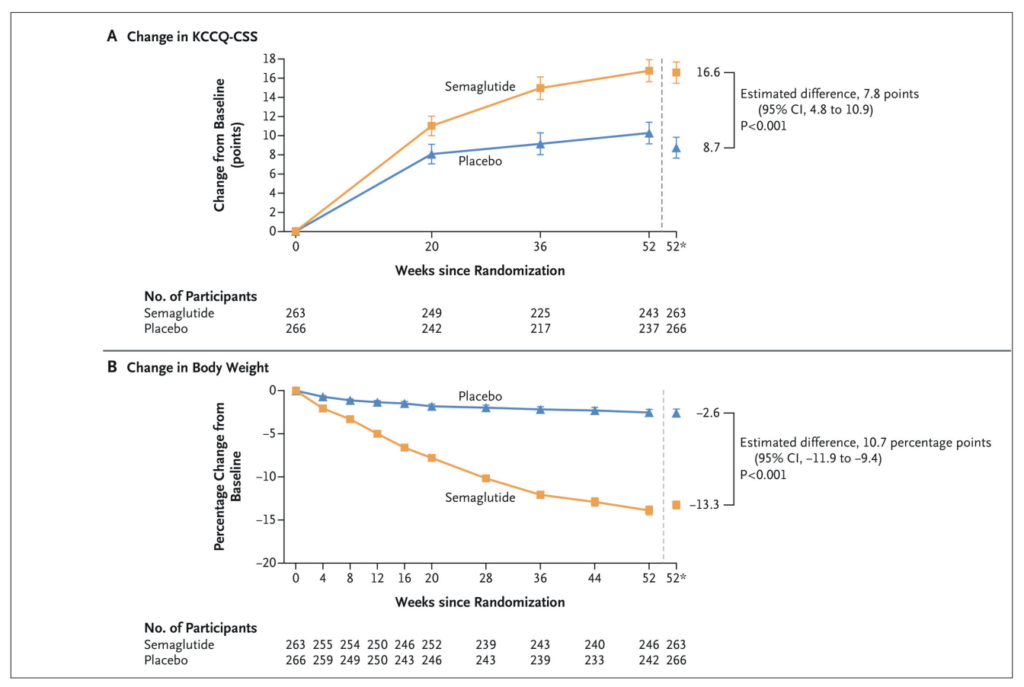
The STEP-HFpEF trial evaluated semaglutide 2.4mg weekly in 529 patients with obesity and HFpEF (NYHA class II–IV, LVEF ≥45%) without diabetes.15 At 52 weeks, semaglutide produced a clinically meaningful improvement in KCCQ-CSS (treatment difference 7.8 points, p<0.001) and a striking 13.3 per cent reduction in body weight versus 2.6 per cent with placebo (p<0.001) (Figure 6).
The companion STEP-HFpEF DM trial showed similar benefits in patients with type 2 diabetes. A pooled analysis by Kosiborod et al incorporating data from SELECT, FLOW, and both STEP-HFpEF trials, demonstrated that semaglutide reduced the composite of cardiovascular death or worsening heart failure (HR 0.69, 95% CI 0.53–0.89, p=0.0045).16
| TRIAL | INTERVENTION | N | DRUG CLASS |
|---|---|---|---|
| CONFIRMATION-HF | Finerenone + SGLT2i vs SoC in hospitalised HF patients | 1,500 | nsMRA + SGLT2i |
| REDEFINE-HF | Early finerenone vs placebo in hospitalised HF (LVEF >40%) | 5,200 | nsMRA |
| BalanceD-HF | Balcinrenone + dapagliflozin vs dapagliflozin in chronic HF | 4,800 | nsMRA + SGLT2i |
| SPIRRIT | Spironolactone vs placebo in stable HFmrEF/HFpEF | 2,000 | sMRA |
| MARITIME HF | Maridebart cafraglutide vs placebo in obese HFpEF | Event- driven |
GLP-1 RA+ |
| EASi-HF | BI 690517 + empagliflozin vs empagliflozin in chronic HF | 6,000 | ASi + SGLT2i |
TABLE 1: The CURRENT HFpEF clinical trial landscape
The SUMMIT Trial: Tirzepatide in HFpEF
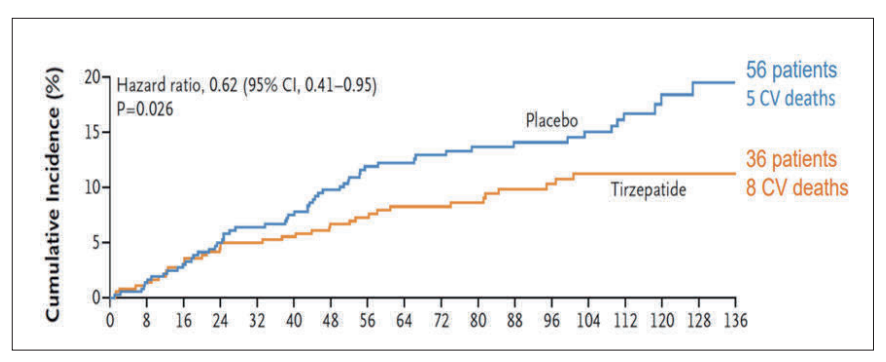
The SUMMIT trial evaluated tirzepatide, a dual GIP/GLP-1 RA, in 731 patients with obesity and stable HFpEF (NYHA class II–IV, LVEF ≥50%).17 Tirzepatide significantly reduced the co-primary endpoint of worsening heart failure or cardiovascular death (HR 0.62, 95% CI 0.41–0.95, p=0.026) (Figure 7).
Notably, while there were numerically more cardiovascular deaths in the tirzepatide arm (8 vs 5), this was more than offset by a substantial reduction in worsening heart failure events (28 vs 51), underscoring that the benefit is driven by heart failure event reduction rather than mortality.
Together with the STEP-HFpEF and STEP-HFpEF DM trials, SUMMIT establishes incretin-based therapies as a transformative strategy for the obese HFpEF phenotype, addressing the root cardiometabolic pathology and producing meaningful improvements in symptoms, weight, and clinical outcomes.
Targeting inflammation: The next frontier
Systemic inflammation plays a central role in the pathogenesis of HFpEF. Pro-inflammatory cytokines, including tumour necrosis factor-alpha (TNF-α), interleukin (IL)-6, IL-1, and IL-18, drive endothelial dysfunction, myocardial fibrosis, cardiomyocyte hypertrophy, and renal injury. The recognition that many HFpEF patients exhibit persistently elevated high-sensitivity C-reactive protein (hsCRP) has led to interest in targeted anti-inflammatory strategies.
The HERMES Trial
The HERMES trial is an ongoing cardiovascular outcomes trial evaluating ziltivekimab, a monoclonal antibody targeting IL-6, in patients with heart failure (HFpEF or HFmrEF) and systemic inflammation.18 The trial is enrolling approximately 5,600 patients with elevated hsCRP (≥2mg/L), NYHA class II–IV, LVEF above 40 per cent, elevated NT-proBNP, and echocardiographic signs of HFpEF or HFmrEF.
Patients are randomised 1:1 to ziltivekimab 15mg subcutaneously once monthly versus placebo, with the primary endpoint being the first occurrence of cardiovascular death, hospitalisation for heart failure, or urgent heart failure visit. The trial is event-driven, targeting 845 first primary events over a follow-up period of up to four years. If positive, HERMES would validate the inflammatory hypothesis of HFpEF and open a wholly new therapeutic pathway.
Ongoing and upcoming HFpEF trials
The HFpEF clinical trial landscape has never been more active. Several major trials are currently underway, exploring novel therapeutic strategies and combinations across the disease spectrum (Table 1).
Of particular note is the SPIRIT-HF trial (spironolactone in HFmrEF/HFpEF), which was recently terminated, highlighting the challenges faced by steroidal MRAs in this population and further underscoring the rationale for non-steroidal agents such as finerenone. The ongoing CONFIRMATION-HF and BalanceD-HF trials are investigating combination strategies with finerenone and SGLT2i, which may define the next standard of care.
Conclusion
HFpEF has transitioned from a therapeutic wasteland to a field of extraordinary innovation. The convergence of SGLT2i, non-steroidal MRAs, and incretin-based therapies has created a multi-pillar treatment paradigm for HFpEF that was unimaginable even five years ago.
Ongoing trials targeting inflammation, combination therapies, and novel metabolic pathways promise to further refine and expand therapeutic options. For clinicians, the imperative is clear – diagnose HFpEF early, initiate evidence-based therapy promptly, and remain attuned to the rapidly evolving evidence base that continues to reshape our approach to this common and consequential condition.
Conflicts of interest: None declared
References
- Vasan RS, Xanthakis V, Lyass A, et al. Epidemiology of left ventricular systolic dysfunction and heart failure in the Framingham Study: An echocardiographic study over three decades. JACC Cardiovasc Imaging. 2018;11(1):1-11. doi:10.1016/j.jcmg.2017.08.007.
- Owan TE, Hodge DO, Herges RM, et al. Trends in prevalence and outcome of heart failure with preserved ejection fraction. N Engl J Med. 2006;355(3):251-259. doi:10.1056/NEJMoa052256.
- McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42(36):3599-3726. doi:10.1093/eurheartj/ehab368.
- Paulus WJ, Tschöpe C. A novel paradigm for heart failure with preserved ejection fraction: Comorbidities drive myocardial dysfunction and remodelling through coronary microvascular endothelial inflammation. J Am Coll Cardiol. 2013;62(4):263-271. doi:10.1016/j.jacc.2013.02.092.
- Reddy YNV, Carter RE, Obokata M, et. A simple, evidence-based approach to help guide diagnosis of heart failure with preserved ejection fraction. Circulation. 2018;138(9):861-870. doi:10.1161/CIRCULATIONAHA.118.034646.
- Pieske B, Tschöpe C, de Boer RA, et al. How to diagnose heart failure with preserved ejection fraction: The HFA-PEFF diagnostic algorithm: A consensus recommendation from the Heart Failure Association (HFA) of the European Society of Cardiology (ESC). Eur Heart J. 2019;40(40):3297-3317. doi:10.1093/eurheartj/ehz641.
- Remme WJ, Swedberg K. Task force for the diagnosis and treatment of chronic heart failure, European Society of Cardiology. Guidelines for the diagnosis and treatment of chronic heart failure. Eur Heart J. 2001;22(17):1527-1560. doi:10.1053/euhj.2001.2783.
- McMurray JJ, Adamopoulos S, Anker SD, et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The task force for the diagnosis and treatment of acute and chronic heart failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2012;33(14):1787-1847. doi:10.1093/eurheartj/ehs104.
- Authors/Task Force Members, McDonagh TA, Metra M, et al. 2023 Focused update of the 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) With the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2024;26(1):5-17. doi:10.1002/ejhf.3024.
- Anker SD, Butler J, Filippatos G, et al. Empagliflozin in heart failure with a preserved ejection fraction. N Engl J Med. 2021;385(16):1451-1461. doi:10.1056/NEJMoa2107038.
- Solomon SD, McMurray JJV, Claggett B, et al. Dapagliflozin in heart failure with mildly reduced or preserved ejection fraction. N Engl J Med. 2022;387(12):1089-1098. doi:10.1056/NEJMoa2206286.
- Pitt B, Pfeffer MA, Assmann SF, et al. Spironolactone for heart failure with preserved ejection fraction. N Engl J Med. 2014;370(15):1383-1392. doi:10.1056/NEJMoa1313731.
- Solomon SD, McMurray JJV, Vaduganathan M, et al. Finerenone in heart failure with mildly reduced or preserved ejection fraction. N Engl J Med. 2024;391(16):1475-1485. doi:10.1056/NEJMoa2407107.
- Kitai T, Kohsaka S, Kato T, et al. JCS/JHFS 2025 Guideline on diagnosis and treatment of heart failure. Circ J. 2025;89(8):1278-1444. doi:10.1253/circj.CJ-25-0002.
- Kosiborod MN, Abildstrøm SZ, Borlaug BA, et al. Semaglutide in patients with heart failure with preserved ejection fraction and obesity. N Engl J Med. 2023;389(12):1069-1084. doi:10.1056/NEJMoa2306963.
- Kosiborod MN, Deanfield J, Pratley R, et al. Semaglutide versus placebo in patients with heart failure and mildly reduced or preserved ejection fraction: A pooled analysis of the SELECT, FLOW, STEP-HFpEF, and STEP-HFpEF DM randomised trials. Lancet. 2024;404(10456):949-961. doi:10.1016/S0140-6736(24)01643-X.
- Packer M, Zile MR, Kramer CM, et al. Tirzepatide for heart failure with preserved ejection fraction and obesity. N Engl J Med. 2025;392(5):427-437. doi:10.1056/NEJMoa2410027.
- Petrie M, Borlaug B, Buchholtz K, et al. HERMES: Effects of ziltivekimab versus placebo on morbidity and mortality in patients with heart failure with mildly reduced or preserved ejection fraction and systemic inflammation. J Card Fail. 2024;30(1):126. doi:10.1016/j.cardfail.2023.10.024.