Women with T2DM have a similarly high risk for pregnancy complications as pregnant women with type 1 diabetes
Summary
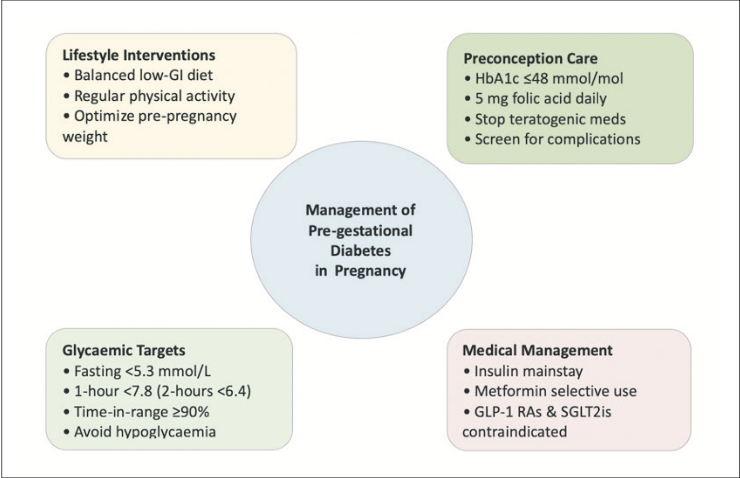
Pre-conception
Provide pre-conception counselling at every visit; aim HbA1c ≤48mmol/mol. Plan a switch to insulin and/or metformin; stop other agents before conception.
Glycaemic targets
Fasting glucose ≤5.3mmol/L, one-hour glucose ≤7.8 or two-hour glucose ≤6.7mmol/L; HbA1c ≤42mmol/mol.
First-line in pregnancy
Basal-bolus insulin; titrate frequently used to achieve target glycaemic range. Requirements rise from ~16 weeks and may fall near term; therefore, close monitoring and frequent dose titration is required.
Metformin
Useful add-on/alternative to insulin; lowers insulin dose and gestational weight gain. Studies have shown that metformin reduces the rate of large for gestational age infants, but signals a higher rate of small for gestational age infants.
GLP-1 RA
Recommendation is to avoid in pregnancy as insufficient human safety data/long-term data and there are animal teratogenicity signals. Advise to stop GLP-1 RA pre-conception and switch to insulin and/or metformin.
SGLT2i
Avoid in pregnancy as there is placental transfer and animal data suggestive of foetal renal injury; no pregnancy efficacy data. Stop pre-conception or immediately on recognition and switch to insulin and/or metformin.
With the global rise in obesity, the prevalence of diabetes in pregnancy is rising. The incidence of type 2 diabetes mellitus (T2DM) in pregnancy is 0.2 per cent. There are significant risks associated with diabetes in pregnancy, which include macrosomia, neonatal hypoglycaemia, neonatal hyperbilirubinaemia, foetal anomalies, spontaneous abortion, and pre-eclampsia. Intrauterine exposure to hyperglycaemia may result in consequences later in life for offspring, including increased incidence of T2DM, obesity, and hypertension.
Pre-conception management
Given the risks, pre-conception counselling (PCC) is essential, and intention for pregnancy should be discussed at every diabetes review for women of childbearing age living with T2DM. Counselling should emphasise optimising glycaemic control before conception, with a target HbA1c ≤48mmol/mol to minimise adverse pregnancy and foetal outcomes. PCC decreases the incidence of adverse pregnancy outcomes such as congenital malformations. Despite the evidence behind PCC, disappointingly, only approximately 20 per cent of individuals living with pre-existing diabetes undergo this counselling.
For women living with T2DM who are planning pregnancy, the American Diabetes Association (ADA) recommends treatment with insulin, as there is very limited safety data on other antidiabetic agents. The UK’s National Institute for Health and Care Excellence guidelines suggest using metformin as an adjunct or alternative to insulin. However, the unintentional exposure to other agents during the first trimester has increased, although it is important to note that it is still rare.
It is universally recommended that folic acid 5mg is commenced prior to conception until 12 weeks gestation. It is also recommended to stop/avoid other teratogenic medications women living with diabetes may be on, for example, ACE (angiotensin-converting enzyme) inhibitors or ARBs (angiotensin II receptor blockers).
It is advised if required for blood pressure control to switch to an alternative like labetalol, methyldopa, diltiazem, etc. It is also recommended to stop statins pre-pregnancy as they are teratogenic.
Risk of congenital malformations in babies born to mothers with diabetes
There is an increased risk of major congenital malformations (MCMs) in babies born to mothers living with T2DM – 5.3 per cent versus 3.7 per cent in the background population. An observational cohort study based in four Nordic countries evaluated periconception exposure to oral hypoglycaemic agents (OHAs) and glucose-like peptide-1 receptor agonists (GLP-1 RAs). This study reported a similar incidence of MCMs following exposure to insulin (7.8%, n=5078) vs other antidiabetic agents: GLP-1 RAs (8.3% n=938), sodium-glucose co-transporter-2 inhibitors (SGLT2i) (7.0% n=335), sulfonylureas (9.7%, n=1362) and dipeptidyl peptidase 4 (DPP-4) inhibitors (6.1%, n=687). Because of this, it is recommended that patients’ diabetes management is optimised pre-conception and that medications are adjusted appropriately to minimise the risk of MCMs that result from either hyperglycaemia or exposure to anti-diabetic agents.
Glycaemic goals in pregnancy
Glycaemic goals during pregnancy are tighter. Fasting, pre-prandial, and post-prandial measurements are recommended to achieve target glucose levels. Targets are as follows: Fasting plasma glucose ≤5.3mmol/L and either one-hour postprandial glucose ≤7.8mmol/L or two-hour postprandial glucose ≤6.7mmol/L. Target HbA1c is lower in the second and third trimester of pregnancy (≤42mmol/mol) as red cell turnover is increased. If the patient is experiencing hypoglycaemia, the target HbA1c can be relaxed to ≤53mmol/mol.
The use of continuous glucose monitoring in T2DM in pregnancy
The most recent Joint Endocrine Society and European Society of Endocrinology guideline advises either continuous glucose monitoring (CGM) or self-monitored blood glucose are acceptable; however, there is no direct evidence to demonstrate CGM results in superior glycaemic control. There is surprisingly limited data in this area. It is known that the use of CGM in T1DM in pregnancy improves neonatal outcomes – such as lower risk of large for gestational age (LGA), fewer admissions to neonatal intensive care, and fewer incidents of neonatal hypoglycaemia – and also reduces adverse pregnancy outcomes. A recent systematic review has concluded that CGM was not associated with a lower rate of LGA infants or pre-eclampsia in T2DM compared to standard-of-care controls. If CGM is used in T2DM in pregnancy, the recommended target time in range is 90 per cent.
Insulin: The preferred agent for treatment
Insulin is the first-line agent for treatment of T2DM in pregnancy as it does not cross the placenta because of its large molecular size. It also has a very well-established safety profile, both maternally and for the infant. Basal-bolus insulin regimes using rapid-acting analogues, such as aspart or lispro, are recommended, with regular titration based on glucose profiles. The physiology of pregnancy necessitates frequent titration of insulin to match changing requirements. From 16 weeks of gestation, insulin resistance increases; this leads to an approximately 5 per cent increase in insulin requirement per week. The ratio of basal to bolus insulin changes, with bolus proportions taking up a greater proportion of the total daily dose of insulin. Towards the final weeks of pregnancy, insulin requirements decline, and this indicates the development of placental insufficiency.
Maternal outcomes for women with T2DM in pregnancy on insulin include improved glycaemic profile and reduced incidence of LGA infants, but there are also increased risks of hypoglycaemia and weight gain. Approximately half of infants born to mothers with pre-existing T2DM are diagnosed and treated for neonatal hypoglycaemia compared to one in five infants of mothers with gestational diabetes. Glycaemic control needs to be optimal intrapartum as hyperglycaemia contributes to neonatal hypoglycaemia.
Approximately half of infants born to mothers with pre- existing T2DM are diagnosed and treated for neonatal hypoglycaemia compared to one in five infants of mothers with gestational diabetes
Insulin requirements often fall during the active stage of labour. When insulin is required during labour, a variable rate infusion titrated as per the local hospital policy is required. In the immediate postpartum period, there is a marked decline in insulin resistance. This is because the placenta has been removed; therefore, there is no longer exposure to placental-mediated hormone production, including human placental lactogen, cortisol, and progesterone. In women who were managed on OHAs pre-pregnancy, insulin should be discontinued immediately post-delivery. In individuals who required insulin therapy pre-pregnancy, it is recommended to reduce the third trimester dose of insulin to 30-40 per cent of the total daily dose, and further reductions may be needed if breastfeeding is commenced. Metformin alone is recommended for women who were on OHAs pre-pregnancy with an A1c ≤7.5 per cent.
Metformin
Metformin has been used in pregnancy since the 1970s. The ADA recommends metformin as a second-line agent in the treatment of diabetes in pregnancy as it crosses the placenta. Metformin is associated with maternal weight loss and less neonatal hypoglycaemia; however, it is often not effective enough as a monotherapy, and the addition of insulin therapy is quite common.
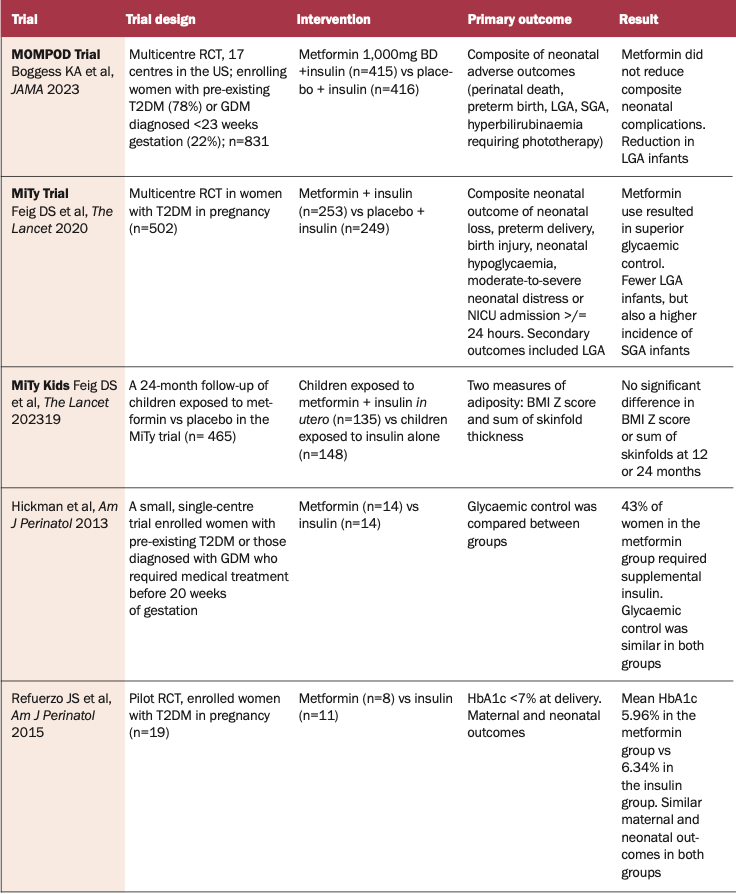
In more recent years, data have been published on efficacy, safety, neonatal, and maternal outcomes on the use of metformin in pregnancy. In 2020, the MiTy trial was published. This was a multicentre, randomised controlled trial looking at neonatal morbidity and mortality in pregnant women with T2DM in pregnancy on metformin in addition to usual care. This trial confirmed that women randomised to the metformin group achieved superior glycaemic control (mean HbA1c 41mmol/mol versus 43.2mmol/mol in the placebo group at 34 weeks of gestation) and required less insulin (1.1 units/kg versus 1.5units/kg in the placebo group; p<0.0001). These women had lower caesarean section rates compared to placebo (53% versus 63%). There were fewer LGA infants (9% in the metformin group versus 15% in the placebo group), but also a higher incidence of small for gestational age (SGA) (13% versus 7%).
In addition to this, there was a 24-month follow-up period of children born to the women enrolled in this trial. The MiTy Kids trial confirmed that there is no significant overall difference in BMI Z score or sum of the skinfolds at 12 months or 24 months old. There was, however, a difference in male weight trajectories in metformin compared to placebo. Male BMI Z score was higher between six months to 24 months in the metformin group.
The MOMPOD trial aligned with the MiTy trial in that there was no significant reduction in composite neonatal morbidity, and it also showed a reduction in LGA infants (>90th percentile) in the metformin-treated group versus placebo (26% versus 36%; OR 0.63).Interestingly, the European guidelines recommend against the use of metformin in individuals already on insulin in an effort to reduce the insulin dose. This suggestion is based on the guideline panel judgement that the benefit of adding metformin to reduce the risk of LGA infants did not outweigh the potential harm of increased incidence of SGA infants.
Sulfonylureas
Sulfonylureas are not recommended in pregnancy. Glyburide crosses the placenta to 50-70 per cent of maternal levels, and there is a higher incidence of neonatal hypoglycaemia and macrosomia when compared to insulin and metformin.
DPP-4 inhibitors
DPP-4 inhibitors are not recommended in pregnancy as there is a lack of pregnancy-specific efficacy and safety data. Ex vivo studies have confirmed sitagliptin can cross the placenta.
Of note, outside of pregnancy, DPP-4i have modest HbA1c reduction. There are alternative efficacious anti-hyperglycaemic medications that we have safety data on in pregnancy.
GLP-1 RA
The current European and American guidelines do not recommend the use of GLP-1 RAs for the treatment of T2DM in pregnancy due to limited safety data. Pre-conception, the discontinuation of GLP-1 RA therapy is recommended. Sudden discontinuation of GLP-1 RAs may result in hyperglycaemia and weight regain; therefore, it is important to plan this appropriately and substitute another anti-hyperglycaemic agent, such as metformin or insulin, if required. Uncontrolled hyperglycaemia can contribute to teratogenicity, and weight gain can affect fertility; therefore, in women planning pregnancy, this transition needs to be monitored very closely. This was suggested by the most recent Joint Endocrine Society and European Society of Endocrinology guideline, as there is a limited evidence database on the risk of exposure to GLP-1 RA during pregnancy.
The concerns regarding these agents in pregnancy are related to adverse outcome reports in animal studies. Animal studies show that there are associated adverse outcomes in offspring, including reduced foetal growth, skeletal and visceral anomalies, and embryonic death.
With the increasing use of GLP-1 RAs for the treatment of obesity, particularly to improve fertility and also the treatment of T2DM, there have been unintentional exposures to GLP-1 RA therapy in early pregnancy. There is extremely limited human data on the safety profile of GLP-1 RA therapy in pregnancy.
Although there are no prospective studies in humans, we have not seen a pattern of congenital malformations in infants. There has been one recent large trial evaluating the safety of GLP-1 RA therapy in early pregnancy. Cesta et al published an observational multinational population-based cohort study of >50,000 women with T2DM. Reassuringly, infants exposed to GLP-1 RA, SGLT2i, DPP4-i and sulfonylureas did not suffer malformations in comparison to those treated with insulin therapy. Because data is limited and there is insufficient evidence regarding long-term offspring outcomes, stillbirth, abortion, neurodevelopment, and maternal glucose control, the European and American guidelines advise to discontinue GLP-1 RA pre-conception.
A Danish population study published in 2024 identified 34 out of 104,422 singleton pregnancies exposed to semaglutide during the first trimester, 17 discontinued in the first trimester, and the remainder continued throughout all trimesters. Semaglutide showed comparable risks of malformations, preterm delivery, LGA, neonatal hypoglycaemia and jaundice compared to pregnancies solely exposed to insulin.
In 2018, there was a case published on an individual who remained on liraglutide for the duration of her pregnancy. The last dose of liraglutide was taken 3.5 hours before delivery via Caesarean section, and a healthy newborn male was delivered with no complications. The concentration of liraglutide was measured in maternal blood and from the umbilical vein, and it was proven that there was no significant transfer of liraglutide to the foetus 3.5 hours after administration.
More recently, in 2024, a case was published on the use of dulaglutide until 34 weeks of gestation in a 42-year-old woman with pre-existing T2DM. She was on atorvastatin, metformin, and dulaglutide. She did not present until 34 weeks of gestation, and at this time, all three agents were immediately stopped. She was commenced on basal bolus insulin therapy. Her pregnancy was complicated by polyhydramnios. An elective Caesarean section was performed at 38 weeks due to breech presentation of the foetus. No congenital malformations or neonatal complications were observed.
We cannot make a conclusion based on single case reports or small observational studies as to the potential risks of GLP-1 RA therapy in pregnancy, but it is important that these cases are reported in the literature to gather more information to make a conclusion regarding the safety of these medications in pregnancy. At present, patients should be counselled that there is insufficient evidence to support safe use of these agents in pregnancy. It is reported in the literature that absorption of the oral contraceptive pill is impaired in those on GLP-1 RA therapy. Therefore, women on GLP-1 RA must be provided with appropriate instruction around this. No adjustment is needed for liraglutide or semaglutide. However, for individuals on tirzepatide, a backup contraceptive is recommended for four weeks after starting or increasing the dose. Non-oral contraception, ie, intra-uterine device or implant, etc, is preferred in women of childbearing age on GLP-1 RA.
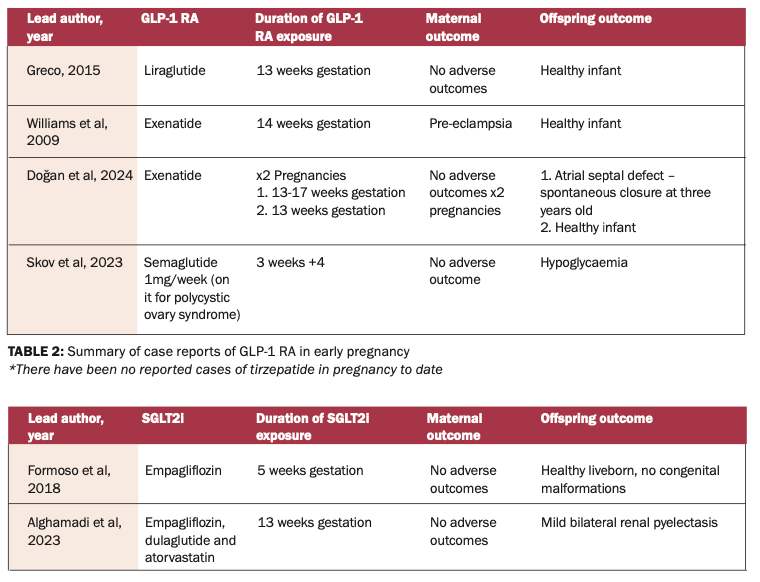
TABLE 3: Summary of case reports of SGLT2i use in pregnancy
SGLT2i
SGLT2i are not recommended in pregnancy. An ex vivo human placental perfusion study has confirmed that SGLT2 inhibitors cross the placenta. Animal data regarding the use of SGLT2i in pregnancy show adverse renal effects such as dilatation of the renal pelvis and tubules of the kidney when used during the second and third trimesters. Therefore, it is advised to avoid SGLT2i in pregnancy. Ideally, they should be stopped pre-conception, and if a patient remains on an SGLT2i, it should be discontinued immediately on recognition of pregnancy.
Conclusion
In conclusion, the current guidelines for treatment of T2DM in pregnancy recommend insulin therapy as the first-line agent. Metformin is recommended as a second-line agent. It has been used in diabetes in pregnancy since the 1970s. However, metformin has a signal for increased incidence of SGA infants, therefore, growth must be monitored closely.
The current guidelines for treatment of T2DM in pregnancy recommend insulin therapy as the first-line agent. Metformin is recommended as a second-line agent
There is limited data regarding the use of newer agents such as GLP-1 RA and SGLT2i. With obesity and T2DM rising rapidly worldwide and greater use of GLP-1 RA therapy, it is likely that we will have more pregnancies with unintended use of these agents. As there are signals of serious congenital malformations in animal studies, it is not possible to perform a clinical trial in this area and real time reporting of observational studies/unintentional exposure and their long-term sequelae should be made available.
References on request









Leave a Reply
You must be logged in to post a comment.