Reference: March 2026 | Issue 3 | Vol 12 | Page 22
Cardiovascular diseases (CVDs) are the leading global killer, causing about 17.9 million deaths in 2019.1 In Europe, CVD remains the top cause of death, accounting for over three million deaths annually.2 The economic burden is also substantial, with costs attributed to health and long-term care, representing 11 per cent of the EU’s total healthcare spending.3
Familial hypercholesterolaemia
Familial hypercholesterolaemia (FH) is a hereditary disorder marked by elevated low-density lipoprotein cholesterol (LDL-C) levels, significantly increasing the risk of CVD.4 Recent estimates suggest that FH affects approximately one in 311 individuals worldwide, making it one of the most common genetic disorders.5 This prevalence suggests that more than 25 million people globally have FH.
In Ireland, it is estimated that around 20,000 people have FH, yet only a small percentage have received a diagnosis.6 Despite its prevalence, FH remains widely underdiagnosed, highlighting the critical need for comprehensive screening programmes to facilitate early detection and treatment.7
Since FH is an irreversible risk factor, prompt detection is essential for initiating appropriate treatment. Although lipid-lowering therapies (LLTs) such as statins and PCSK9 inhibitors are available, studies indicate that many patients with FH fail to achieve optimal LDL-C reduction.8
This systematic review (SR) examines the effectiveness of LLT in lowering LDL-C levels in women with FH. It also evaluates whether current treatment strategies adequately reduce CVD risk in this population. Women with FH tend to develop atherosclerotic CVD (ASCVD) about seven to 20 years later than men with FH.9
If FH is left untreated, an estimated 30 per cent of women will experience a heart attack by the age of 60 years (versus 8 per cent in the general population), compared to men by the age of 50.10 CVDs remain the leading cause of death among women, particularly due to FH, which increases this risk.11,12 However, there is a significant gap in knowledge regarding the management of CVD risk in women with FH.
Primary care practitioners play a crucial role in educating patients about the risk factors for CVD by promoting adherence to LLTs and overseeing their overall cardiovascular health.13 Monitoring LDL-C levels is essential for diagnosing FH, evaluating treatment effectiveness, and maintaining target levels to lower cardiovascular risk. Women with FH should strive for an LDL-C level below 2.6mmol/L or a minimum reduction of 50 per cent from their baseline levels.14
Women with FH and high LDL-C levels often experience delays in diagnosis, limited access to LLTs, and treatment gaps during pregnancy and breastfeeding.15 Research indicates that FH remains significantly underdiagnosed in many countries, with disparities in healthcare infrastructure affecting access to screening and treatment.4,16 While some countries, such as the Netherlands and the United Kingdom, have well-established FH screening programmes, others struggle with insufficient resources and awareness, leading to missed diagnoses and suboptimal treatment.
In Ireland, the management of FH in primary care settings needs to be improved, with limited access to genetic screening in some regions.17 A lack of resources can hinder general practitioners’ ability to identify at-risk patients, resulting in inadequate care for those with FH.12,18 The above issues are important to explore, particularly for women who face the dual burden of managing both FH and elevated CVD risk.19
Despite the established link between FH and CVD, research on the effectiveness of LLT, specifically in women with FH, remains limited. A preliminary search was completed across significant databases, including PubMed, CINAHL, EMBASE, Scopus, and Cochrane, which did not identify a completed SR addressing this question.
Given the existing knowledge gap, this review is essential to evaluating whether current lipid therapies adequately reduce LDL-C levels and prevent CVD in this high-risk population. This SR aims to inform clinical practice and guide future research on optimising FH management in women by synthesising available evidence.
Methods
Design: The SR includes various studies, including randomised controlled trials (RCTs), cohort studies, and observational studies. These different designs contribute to variability in the quality and reliability of evidence. These studies were searched using the Cochrane Handbook for Systematic Reviews.20
Aim and research question: This SR examined the impact of LLT on the incidence of CVD among women with FH. The PICO framework assisted in developing the research question and formulating the inclusion and exclusion criteria.
PICO FRAMEWORK
Population: FH in women
Intervention: LLTs
Comparison: No LLT, placebo, or other therapies (lifestyle changes)
Outcome: Incidence of CVD.
OUTCOMES MEASURED
Primary outcome: To assess the impact of LLT on the incidence of CVD in women with FH
Secondary outcomes:
- Compare the development of CVD with and without therapy
- Comparison of the effectiveness of different LLTs
- Sex-specific differences in LLT outcomes
- Adherence to LLT and its influence on CVD risk
- Delayed diagnosis and sex difference in FH prevalence.
Search strategy
The SR included studies that met the following criteria.
Inclusion criteria:
- Studies that include both men and women may be considered if data for women can be extracted separately
- Studies evaluating the effects of LLT by LDL-C measurements and CVD outcomes
- Study design: RCTs, retrospective analyses, observational studies, cohort studies, or case-control studies
- Studies published in English.
Exclusion criteria:
- Research involving children
- Investigations lacking LDL-C reduction and cardiovascular outcome measurements
- Study types including case reports, series, editorials, and animal studies.
Search methods
The following electronic databases were searched: Web of Science (2015 to January 2025); CINAHL (2015 to January 2025); PubMed (2015 to January 2025); EMBASE (2015 to January 2025).
A database search was conducted to identify relevant articles, assess their eligibility, and select those that met the predefined inclusion criteria.21 This search was conducted across four electronic databases: EMBASE, CINAHL, PubMed, and Web of Science, all of which are accessible via the Royal College of Surgeons in Ireland (RCSI) library. These databases were chosen for their extensive coverage of medical and health literature, ensuring a broad selection of relevant studies.22,23
The search employed keywords following the PICO framework and MeSH terms to refine and clarify the search: Familial hypercholesterolaemia; Lipid-lowering therapies; women; cardiovascular disease; female; cardiovascular mortality; arteriolosclerosis; cardiovascular disease; incidence; statins; low-density lipoprotein; high cholesterol; coronary heart disease; reducing cholesterol. Boolean operators (‘OR’ and ‘AND’) were used to combine keywords effectively.
Filters were applied to refine the search in line with the inclusion criteria.24 To ensure thoroughness, additional relevant terms were explored using ‘OR’. The most pertinent results were reviewed to identify potential synonyms that could enhance the search.22
Grey literature was also considered, including government reports, health service documents, theses, dissertations, conference papers, and clinical trial registers. Sources such as GreyNet International and OpenGrey were utilised to identify unpublished or non-peer-reviewed studies that might offer supplementary insights.25,26
In addition to database searches, reference lists from studies identified during the initial search were reviewed, which yielded relevant information and data.27 DynaMed was also consulted to obtain the latest guidelines for managing FH.
| AUTHOR | COUNTRY | SAMPLE | PRIMARY OUTCOME | SECONDARY OUTCOME |
STUDY TYPE | EBL SCORE |
|---|---|---|---|---|---|---|
| Alonso et al (2021) |
Spain | 433 (47% female) |
Efficacy of PCSK9 (statin) | CVD risk reduction | Long-term prospective study |
92.86% (valid) |
| Duell et al (2019) |
US | 1,900 patients (60% female) |
Combination therapies | Significant risk reduction in women |
Longitudinal observational study |
80% (valid) |
| Humphries et al (2019) |
UK | 2929 study participants (51% female) |
Statins | Long-term LDL reduction, reduced MI risk |
Cohort study | 76% (valid) |
| Raai et al (2024) |
Multicentred study in eight countries |
56 study subjects (60.7% women) |
Statin (inclisiran) treating FH |
Significant PCSK9 reduction, fewer CVD cases |
Randomised controlled trial |
79.30% (valid) |
| Toell et al (2018) |
Austria | 1,054 patients (46.6% female) |
Prevalence of FH FH in premature stroke/TIA Statin treatment |
Screening for FH LDL-target achievement |
Observational and cross-sectional design |
82.76% (valid) |
| Zamora et al (2023) |
Spain | 2,554,644 (52.6% female) |
Women with FH LLT intensity and adherence |
Reduced CVD incidence |
Retrospective analysis |
76% (valid) |
TABLE 1: Characteristics of included studies
Data extraction
A data extraction template was used to input key data relevant to the question. A table was constructed to outline the characteristics of each study (Table 1). No data transformations were performed. Where datasets were incomplete, values were excluded from synthesis and the issue was noted.
Data analysis and synthesis
This SR employed a narrative synthesis to interpret findings across studies with heterogeneous designs and outcomes. While quantitative data on LDL-C reduction and cardiovascular event rates were reviewed, the variability in study methods and populations limited the feasibility of a purely statistical synthesis.
The narrative approach proved valuable in capturing essential insights that might otherwise be overlooked through quantitative analysis alone. It explored gender-specific themes that are particularly relevant to women with FH. In addition, subgroup patterns were considered to investigate potential modifiers of treatment effects based on age, type of therapy, and adherence levels.
Quality appraisal
Quality appraisal of included studies was conducted using the Evidence-Based Librarianship (EBL) tool, which assessed search results and identified relevant studies to mitigate publication bias.33 The EBL tool employs a structured scoring system that evaluates studies across four key domains: Population, data collection, study design, and results analysis.34
Each domain was assigned a score based on predefined criteria, allowing for an objective assessment of study rigour. The total EBL score was calculated by summing the domain scores. The higher the score, the stronger the quality. Studies above the set threshold were deemed suitable or included, while those with lower scores were excluded. This approach minimised bias, ensuring that only well-conducted studies contributed to the SR, thereby enhancing validity.33
Results
The SR analysed a total of 743 articles based on the inclusion criteria. A date range of the past 10 years was applied to focus on recent studies. The initial database searches identified 743 articles, which were reduced to 564 after eliminating 179 duplicates.
Screening of abstracts led to the exclusion of 311 articles, while a full-text review eliminated 157 for insufficient data on women with FH and 81 that failed to meet the inclusion criteria. Ultimately, 13 studies were adequate and aligned with the inclusion criteria. After using the EBL tool to assess the validity of the studies, six articles were identified as of high quality.
Study designs
Each of the studies used different methodologies to explore various aspects of LLT and CVD outcomes, providing a comprehensive understanding of the topic from different research perspectives. These studies generally followed quantitative methods, focusing on clinical outcomes like LDL-C reduction, CVD risk, and treatment adherence.
For example, three studies utilised observational designs,14,32,36 while two conducted longitudinal cohort studies to evaluate treatment effectiveness over time.3,29 One study utilised an RCT design, offering experimental evidence on the efficacy of LLT.31 No qualitative methods were used in the included studies.
Geographical location
Six studies met the inclusion criteria for various countries worldwide. The studies were conducted in the United States,29 Spain,14,18 United Kingdom,30 Austria,32 and multiple countries.31 This global diversity offers valuable insights into the varying approaches to managing FH across different healthcare systems and populations.
| STUDY | TOTAL PARTICIPANTS |
FEMALE PARTICIPANTS (N) |
PERCENTAGE FEMALE (%) |
|---|---|---|---|
| Alonso et al (2021) | 433 | 204 | 47% |
| Duell et al (2019) | 1900 | 1140 | 60% |
| Humphries et al (2019) | 2929 | 1494 | 51% |
| Raai et al (2024) | 56 | 31 | 60.7% |
| Toell et al (2018) | 1054 | 491 | 46.6% |
| Zamora et al (2023) | 2,554,644 | 1,256,800 | 49.2% |
TABLE 2: Female study participants
Participants and sample size
Study participants were adults diagnosed with FH. Many studies included sub-data on women with FH and their response to LLT. The focus on assessing CVD risk and the impact of specific treatments is a common theme in most studies. The review analysed data from six high-quality studies, encompassing a total of 2,561,016 participants, and providing a broad representation of FH populations. The smallest study31 included 56 participants, whereas the largest18 analysed 2,554,644 individuals. Table 2 provides insights into female participants included in each study.
As illustrated in Table 3, high-intensity statins and PCSK9 inhibitors were the most commonly used LLTs, with reported LDL-C reductions ranging from 50 per cent to over 60 per cent in several studies.14,29
Despite therapeutic efficacy, LDL-C target achievement remained suboptimal in some studies, particularly among women with homozygous FH (HoFH).31 Treatment duration and follow-up varied considerably, which may have contributed to the heterogeneity of outcomes. Notably, three studies14,29,30 reported a clear association between LLT use and reduced CVD mortality among female FH patients.
Primary outcomes
Women with FH derive substantial CVD risk reductions from LLT. Statins-based treatment dramatically lowers coronary risk in women with FH compared to historical outcomes without therapy. Untreated FH carries a very high lifetime risk, with approximately 30 per cent of women with FH experiencing a coronary event by the age of 60.18
The included studies indicate that women initiating LLT before CVD onset achieved a 76 per cent relative risk reduction in coronary events. The types of outcomes reported across these studies, such as CVD incidence, mortality, and LLT effectiveness, are summarised in Table 4.
Data from the SAFEHEART registry demonstrated that PCSK9 inhibitors significantly reduced LDL-C levels by approximately 58-60 per cent, with about 67 per cent of patients achieving LDL-C targets, correlating with a significant reduction in the incidence of cardiovascular events.14
Combination therapies involving statins and PCSK9 inhibitors are highly effective in modifying long-term risk. Conversely, the efficacy of inclisiran appeared limited in individuals with HoFH, showing minimal LDL-C reduction and limited improvement in CVD risk.31
The Simon Broome registry, summarised by Humphries et al,30 reported decreased coronary heart disease mortality among women receiving LLT, especially long-term statin therapy; however, increased mortality persisted in patients with severe FH despite treatment. The CASCADE FH registry data revealed that high-intensity statins and combination therapy regimens led to more significant LDL-C reductions and fewer cardiovascular events.12 A
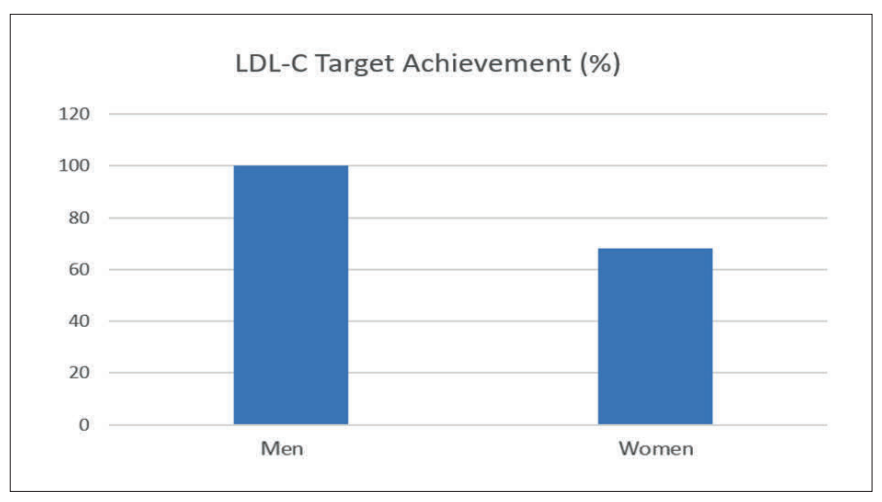
sex-specific disparity was identified in one study, with women being 32 per cent less likely to achieve LDL-C targets compared to men, primarily due to lower adherence and delayed diagnosis.18 Additionally, untreated women with FH experienced ASCVD events around 20 years earlier than untreated men with FH.32
| STUDY | WOMEN INCLUDED (N,%) |
FH TYPES | LLTS USED | DOSE & REGIMEN |
|---|---|---|---|---|
| Alonso et al (2021) | 204 (47%) | HeFH | Atorvastatin, Evolocumab |
High dose statin and PCSK9i |
| Duell et al (2019) | 1140 (60%) | Mixed HeFH/HoFH |
Rosuvastatin+ Ezetimibe Alirocumab |
Max Statin +/- ezetimibe/PCSK9i |
| Humphries et al (2019) |
1494 (51%) | HeFH | Simvastatin, Atorvastatin |
Long-term standard statin |
| Raai et al (2024) | 31 (60.7%) | HoFH | Inclisiran | Inclisiran 284mg biannually |
| Toell et al (2018) | 491 (46.6%) | HeFH | Simvastatin | Standard dose statins |
| Zamora et al (2023) | 1,256,800 (49.2%) | HeFH | Various statins inc |
| TREATMENT DURATION |
FOLLOW UP DURATION |
LDL REDUCTION (%) |
OTHER LIPIDS AFFECTED | TARGET ACHIEVEMENT |
MORTALITY REDUCTION |
|---|---|---|---|---|---|
| >5 years | 6 years | 58-60% | Decrease in total-C and ApoB | 67% | Yes |
| >3 Years | 3 Years | 54% | Decrease in non-HDL-C | 58% | Yes |
| 10+ years | 20 years | 50% | Decrease in triglycerides | 65% | Yes |
| 1 year | 12 months | 10% | Minimal changes | <20% | No |
| N/A | Not specified | 40-50% | Not reported | Not measured | Not measured |
| 5 years | 5 years | Varied | Decrease in non-HDL-C and lp(a) | Men 32% more likely | Sex disparity noted |
TABLE 3: Summary of study characteristics, treatment regimens, and outcomes in women with FH
| AUTHORS | PRIMARY OUTCOME |
SECONDARY OUTCOMES | |
|---|---|---|---|
| IMPACT OF LLT ON CVD |
REDUCTION IN CVD-RELATED MORTALITY |
EFFECTIVENESS OF DIFFERENT LLTS |
|
| Alonso et al (2021) | ✔ | ✔ | ✔ |
| Duell et al (2019) | ✔ | N/M | ✔ |
| Humphries et al (2019) | ✔ | ✔ | ✔ |
| Raai et al (2024) | ✔ | N/M | ✔ |
| Toell et al (2018) | ✔ | N/M | N/M |
| Zamora et al (2023) | ✔ | N/M | N/M |
TABLE 4: Outcomes measured
| THERAPY | LDL-C REDUCTION (%) | CVD EVENT REDUCTION (%) |
|---|---|---|
| Statins | 50% | 40-80% |
| Ezetimibe | 15% | 10-25% |
| PCSK9 inhibitors | 54-61% | 60% |
| Inclisiran | 10% (HoFH cases) | Minimal in HoFH |
TABLE 5: Comparison of LDL-C and CVD event reductions by the LLTs
Secondary outcomes
A consistent pattern emerged across the included studies, which showed that women who received LLT, particularly high-intensity statins and PCSK9 inhibitors, experienced substantially lower rates of adverse cardiovascular outcomes than those who remained untreated or under-treated. Patients receiving LLT, particularly those on a PCSK9 inhibitor, showed a significant reduction in LDL-C levels, with a median drop of 58 mg/dL.14,29 In contrast, untreated women with FH faced a nearly six-fold increase in major adverse cardiovascular events, especially among those with pre-existing ASCVD.18,29,30
Several studies included in this review indicated that women who initiated treatment early and achieved sustained LDL-C reduction experienced a lower incidence of coronary events compared to their untreated counterparts.18,29,30 These findings are consistent with the outcomes reported across the studies, as detailed in Table 3.
Comparison of the effectiveness of different LLTs
Statins remain the first-line treatment for women with FH, reducing LDL-C by approximately 50 per cent. High-intensity statin therapy alone was associated with 40-80 per cent relative risk reduction in CV events.29,30 Where statins were insufficient, adjunct therapies such as ezetimibe and PCSK9 inhibitors contributed to additional LDL-C reductions. Duell et al29 observed a 15-25 per cent LDL-C reduction with ezetimibe, especially in secondary prevention contexts.
PCSK9 inhibitors achieved 54-61 per cent LDL-C reduction, mainly when used in conjunction with maximised statin and ezetimibe therapy.14 SAFEHEART registry data showed that 67 per cent of patients on PCSK9 inhibitors reached LDL-C targets. In contrast, inclisiran showed minimal LDL-C reduction in patients with HoFH. However, the SR also revealed limitations in therapeutic response, particularly in patients with HoFH, likely due to impaired LDL receptor function.31 A summary of the comparative effectiveness of these therapies in terms of LDL-C and CVD event reduction is provided in Table 5.
Sex-specific differences in LLT outcomes
Real-world data show that women often receive lower treatment intensities, experience delayed therapy initiation, and have a lower likelihood of attaining recommended LDL-C targets.29,18 Women are approximately 32 per cent less likely than men to reach LDL-C targets.18 Factors such as provider decision-making, pregnancy-related concerns, and healthcare access differences have been identified as contributing to the underutilisation of optimal therapy in female FH patients, as seen in Figure 1.
Adherence to LLT and its influence on CVD risk
Studies consistently reported lower medication adherence rates in women compared to men, with a direct impact on LDL-C reduction and CVD event prevention.18 Barriers included socioeconomic factors, psychological concerns, and adverse effects such as myalgias and statin intolerance.
Delayed diagnosis and sex difference in FH prevalence
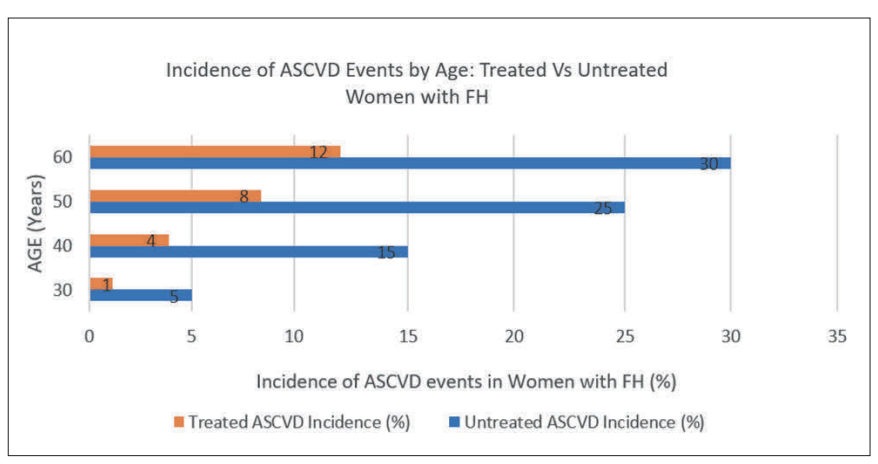
Women with FH were often diagnosed later than men, particularly following stroke or transient ischaemic attack (TIA) events.32 This delay resulted in a more prolonged exposure to elevated LDL-C levels and a higher incidence of premature ASCVD,18 as shown in Figure 2.
Data presented in Figure 2 illustrate that untreated women with FH had an ASCVD incidence of approximately 30 per cent by the age of 60. In contrast, those receiving LLT had substantially lower event rates (12%) by the same age.14,30,32 This represents a 60 per cent relative risk reduction, emphasising the life-prolonging potential of early and intensive LLT in women.
Results of quality appraisal
The EBL scores for these studies ranged from 76-93 per cent, indicating methodological rigour. The majority of the studies achieved high EBL scores (above 75%), demonstrating methodological rigour and the inclusion of relevant and reliable data. The studies had a variety of designs, including long-term prospective studies, longitudinal observational studies, randomised control trials (RCTs), cohort studies, and retrospective analyses. Each study provided valuable insight into the impact of LLT in reducing CVD risk in individuals with FH, as shown in Table 6.
Discussion
This SR shows that LLT, especially statins and PCSK9 inhibitors, significantly lower CVD incidence among women with FH. The reviewed studies consistently reported notable decreases in LDL-C, achieving about a 50 per cent reduction, with PCSK9 inhibitors providing an additional 60 per cent reduction when used with other treatments.30,37 Observational evidence indicates that women who receive early and ongoing LLT can experience up to an 80 per cent relative risk reduction in CVD events compared to those who remain untreated.10,14,37,38
The review also highlighted gender differences in treatment practices and outcomes. Research by Duell et al29 and Zamora et al18 indicates that women with FH receive less intensive LLTs and are less likely to meet LDL-C targets than men. Later diagnoses and lower adherence rates among women further intensify this disparity.
| THE VALIDITY OF INCLUDED STUDIES | |||||
|---|---|---|---|---|---|
| AUTHOR | CATEGORY RESULT % | OVERALL RESULT |
|||
| POPULATION | DATA COLLECTION |
STUDY DESIGN | RESULTS | ||
| Alonso et al (2021) | Validity calculation: 87.5% |
Validity calculation: 100% |
Validity calculation: 100% |
Validity calculation: 100% |
92.86% (valid) |
| Duell et al (2019) | Validity calculation: 66.67% |
Validity calculation: 62.5% |
Validity calculation: 100% |
Validity calculation: 100% |
80% (valid) |
| Humphries et al (2019) |
Validity calculation: 66.67% |
Validity calculation: 62.5% |
Validity calculation: 100% |
Validity calculation: 83.3% |
76% (valid) |
| Raai et al (2024) | Validity calculation: 70% |
Validity calculation: 62.5% |
Validity calculation: 100% |
Validity calculation: 100% |
79.3% (valid) |
| Toell et al (2018) | Validity calculation: 66.67% |
Validity calculation: 62.5% |
Validity calculation: 80% |
Validity calculation: 83.33% |
82.76% (valid) |
| Zamora et al (2023) | Validity calculation: 50% |
Validity calculation: 75% |
Validity calculation: 100% |
Validity calculation: 80% |
|
TABLE 6: Study validity
The studies highlight obstacles including worries about fertility and pregnancy, restricted healthcare access, and socio-cultural influences such as caregiving roles.22,39 Statins were shown to decrease major vascular events by approximately 20 per cent per millimoles per litre LDL-C reduction in men and about 16 per cent in women.12,37,40,41 Likewise, PCSK9 inhibitor trials included in this review demonstrated consistent risk reductions for both men and women, reinforcing their value as monotherapy and adjunctive therapy in high-risk groups.11,14
New treatments, such as inclisiran, show potential for effective long-term LDL-C management, particularly due to their biannual administration, which could enhance patient adherence.
Nonetheless, those with HoFH showed limited effectiveness, indicating a need for personalised treatment strategies.31 This SR highlights that women with FH who receive proper treatment experience relative reductions in CVD similar to those of men.
However, treatment disparities in the real world continue to obstruct optimal outcomes. These disparities stem from factors like clinical decision-making, the absence of sex-specific guidelines, and practical challenges, including medication costs and limited access to specialised care.39,42,43
However, despite the established efficacy of these treatments, evidence reveals that women with FH frequently do not fully benefit from available therapies due to systematic undertreatment and unique barriers.
Studies from Europe and the United States consistently report that women are prescribed less intensive LLT and are significantly less likely to achieve lipid targets than men.36,38,44 Reasons cited included outdated beliefs about female CVD risk, safety concerns related to reproductive health, and clinical misperception of risk urgency.18 The result is prolonged cumulative LDL-C exposure and increased atherosclerotic burden, often becoming clinically apparent in later life.30
Importantly, the included studies affirm that adequately treated women with FH derive comparable lifetime benefits from LLT. Although risk reduction appears less pronounced in premenopausal women, the shift of risk following menopause underscores the necessity for early initiation and long-term therapy maintenance.29
The review and supporting literature suggest that sex-based differences in adherence, access, and risk perception contribute more to outcome discrepancies than pharmacologic efficacy. Barriers such as the expense of advanced treatments like PCSK9 inhibitors and inclisiran, as well as gaps in insurance coverage in various countries and interruptions in care, disproportionately affect women. This is due to life-course factors, such as pregnancy and limited access to specialists in this field.11
Recent real-world data further support the efficacy of LLTs in women with FH. A study by Bosco et al,45 which included 49 per cent female participants, demonstrated that inclisiran effectively reduces LDL-C in hypercholesterolaemic individuals in routine clinical practice. This strengthens the generalisability of LLT effectiveness to real-world female populations.
Despite the benefits, treatment disparities persist. Statin intolerance, which disproportionately affects women, may partly explain lower adherence and intensity of LLT in this group. Casula et al24 highlighted a higher prevalence of statin intolerance among hypercholesterolaemic women compared to men, suggesting a need for better-tailored therapeutic strategies.
Moreover, emerging evidence suggests that biological differences may influence pharmacogenetic responses. A study by Bosco et al46 investigated sex-specific genetic variants associated with poor statin adherence, particularly among women. These findings underscore the importance of considering genetic and sex-specific factors in optimising long-term LLT adherence and CVD outcomes in women with FH.
The review emphasises the urgent need to close the women’s FH care gap. Clinicians are advised to initiate high-intensity statin therapy as early as possible in female patients, reflecting current guidelines that emphasise the importance of early lipid control to prevent vascular damage.29,30,42 PCSK9 inhibitors also provide an effective alternative or adjunctive treatment for women who cannot reach LDL targets or experience statin intolerance.14,42,44
Limitations
This review identified several limitations. Due to considerable heterogeneity in study design, participant populations, interventions, measured outcomes, and reporting standards, a meta-synthesis was not feasible. As such, a narrative synthesis was used to integrate findings. The included studies varied in sample size, treatment types, follow-up durations, and population subgroups, making direct statistical pooling inappropriate.
These studies highlight the transformative impact of LLTs on reducing the burden of cardiovascular procedures and events, providing clear guidance for future clinical practice. Key messages include the timely initiation of therapy, optimal use of combination therapies, and the implementation of sustained long-term prevention strategies, ultimately improving patient outcomes and reducing healthcare system burdens.38,44
A notable limitation of this review is the reliance on predominantly observational data, given the scarcity of explicit RCTs focused on women with FH. Although the consistency of evidence strongly supports LDL-C reduction benefits, the precise magnitude of risk reduction in women with FH depends heavily on observational estimates, which could be influenced by biases such as healthy adherence effects of residual confounders.
Nevertheless, consistent findings across multiple observational cohorts, including the Simon Broome Register, CASCADE-FH, and the Spanish registry, provide substantial credibility to these conclusions.14,29,30 This review did not formally assess reporting bias or certainty of evidence using tools such as the GRADE framework, which may limit the strength of inferences drawn from observational data.
Future research and implications for practice
Future research should focus on conducting high-quality, sex-specific RCTs to evaluate the efficacy and safety of LLTs in women with FH. These trials should explore the impact of hormonal status (eg, menopause, pregnancy), adherence challenges, and pharmacodynamic differences in treatment response. More granular, sex-disaggregated data are needed across all phases of research to support precision medicine approaches to FH management.
There is also a need to develop and test target interventions that address adherence barriers unique to women, such as effective management, reproductive considerations, and caregiver burden. Health services research should investigate models of care that integrate CVD risk screening into routine reproductive and primary care settings.
For clinical practice, these findings reinforce the urgency of initiating LLT early in women and sustaining treatment throughout the life course. Clinicians should adopt a proactive approach to address treatment hesitance, provide education about CVD risk in women, and ensure equitable access to advanced therapies. Enhanced guidelines implementation and multidisciplinary management models involving lipid specialists, primary care providers, and women’s health practitioners will likely improve outcomes for this high-risk population.
Conclusion
This SR highlights that women with FH face a significantly elevated risk of CVD, effectively mitigated by appropriate LLT such as statins, ezetimibe, PCSK9 inhibitors, and inclisiran. These therapies significantly reduce LDL-C and dramatically lower the incidence of cardiovascular events when optimally applied. However, persistent underdiagnosis, undertreatment, and adherence challenges influenced by misconceptions, pregnancy concerns, and healthcare biases create a substantial gender gap in outcomes.
Addressing these barriers through early FH identification in women, aggressive guideline-driven therapy initiation, effective pregnancy management, and equitable access to advanced treatments is essential. Prioritising these areas enables healthcare providers to maximise the therapeutic potential of LLTs, significantly lowering CVD risk in women with FH and ultimately aligning their cardiovascular outcomes.
References
- World Health Organisation. Cardiovascular diseases. Available at: www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)
- Timmis A, Aboyans V, Vardas P, et al. European Society of Cardiology: The 2023 Atlas of Cardiovascular Disease Statistics. Eur Heart J. 2024;45(38):4019-4062. doi:10.1093/eurheartj/ehae466.
- Luengo-Fernandez R, Walli-Attaei M, Gray A, et al. Economic burden of cardiovascular diseases in the European Union: A population-based cost study. Eur Heart J. 2023;44(45):4752-4767. doi:10.1093/eurheartj/ehad583.
- Nordestgaard BG, Chapman MJ, Humphries SE, et al. Familial hypercholesterolaemia is underdiagnosed and undertreated in the general population: Guidance for clinicians to prevent coronary heart disease – consensus statement of the European Atherosclerosis Society. Eur Heart J. 2013;34(45):3478-90a. doi:10.1093/eurheartj/eht273.
- Hu P, Dharmayat KI, Stevens CAT, et al. Prevalence of familial hypercholesterolemia among the general population and patients with atherosclerotic cardiovascular disease: A systematic review and meta-analysis. Circulation. 2020;141(22):1742-1759. doi:10.1161/CIRCULATIONAHA.119.044795.
- Morris T, Harding E, Handcock M. Advancing a prevention agenda for cardiovascular care in Ireland: Position paper. The Health Policy Partnership; 2023.
- Banda JM, Sarraju A, Abbasi F, et al. Finding missed cases of familial hypercholesterolemia in health systems using machine learning. NPJ Digit Med. 2019;2:23. doi:10.1038/s41746-019-0101-5.
- Vallejo-Vaz AJ, Kondapally Seshasai SR, Cole D, et al. Familial hypercholesterolaemia: A global call to arms. Atherosclerosis. 2015;243(1):257-259. doi:10.1016/j.atherosclerosis.2015.09.021.
- Klevmoen M, Mulder JWCM, Roeters van Lennep JE, Holven KB. Sex differences in familial hypercholesterolemia. Curr Atheroscler Rep. 2023;25(11):861-868. doi:10.1007/s11883-023-
01155-6. - Centres for Disease Control and Prevention (CDC). About familial hypercholesterolemia. Available at: www.cdc.gov/heart-disease-family-history/about/about-familial-hypercholesterolemia.html
- 11.Agarwala A, Deych E, Jones LK, et al. Sex-related differences in premature cardiovascular disease in familial hypercholesterolaemia. J Clin Lipidol. 2023;17(1):150-156. doi:10.1016/j.jacl.2022.11.009.
- Durand A, Morgan CL, Tinsley S, et al. Familial hypercholesterolaemia in UK primary care: A Clinical Practice Research Datalink study of an under-recognised condition. Br J Gen Pract. 2024;74(740):e174-e182. doi:10.3399/BJGP.2023.0010.
- Lizcano-Álvarez Á, Carretero-Julián L, Talavera-Saez A, et al. Intensive nurse-led follow-up in primary care to improve self-management and compliance behaviour after myocardial infarction. Nurs Open. 2023;10(8):5211-5224. doi:10.1002/nop2.1758.
- Alonso R, Pérez de Isla L, Muñiz-Grijalvo O, et al. Familial hypercholesterolaemia diagnosis and management. Eur Cardiol Rev 2018; 13(1): 14-20.
- Roeters van Lennep JE, Tokgözoğlu LS, Badimon L, et al. Women, lipids, and atherosclerotic cardiovascular disease: A call to action from the European Atherosclerosis Society. Eur Heart J. 2023;44(39):4157-4173. doi:10.1093/eurheartj/ehad472.
- Beheshti SO, Madsen CM, Varbo A, Nordestgaard BG. Worldwide prevalence of familial hypercholesterolaemia: Meta-analyses of 11 million subjects. J Am Coll Cardiol. 2020;75(20):2553-2566. doi:10.1016/j.jacc.2020.03.057.
- Agar R, Prendergast M, Maher V. Evaluation of lipid services in the Republic of Ireland. Ir J Med Sci. 2020;189(3):925-931. doi:10.1007/s11845-020-02187-6.
- 18.Zamora A, Ramos R, Comas-Cufi M, et al. Women with familial hypercholesterolaemia phenotype are undertreated and poorly controlled compared to men. Sci Rep. 2023;13(1):1492. doi:10.1038/s41598-023-27963-z.
- Gonçalves C, Moreira H, Santos R. Systematic review of mediterranean diet interventions in menopausal women. AIMS Public Health. 2024;11(1):110-129. doi:10.3934/publichealth.2024005.
- Higgins JP, Altman DG, Gøtzsche PC, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi:10.1136/bmj.d5928.
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009; 6(7): e1000097.
- Bramer WM, de Jonge GB, Rethlefsen ML, Mast F, Kleijnen J. A systematic approach to searching: An efficient and complete method to develop literature searches. J Med Libr Assoc. 2018;106(4):531-541. doi:10.5195/jmla.2018.283.
- Hartling L, Featherstone R, Nuspl M, et al. The contribution of databases to the results of systematic reviews: A cross-sectional study. BMC Med Res Methodol. 2016;16(1):127. doi:10.1186/s12874-016-0232-1.
- Casula M, Gazzotti M, Bonaiti F, et al. Reported muscle symptoms during statin treatment amongst Italian dyslipidaemic patients in the real-life setting: The PROSISA Study. J Intern Med. 2021;290(1):116-128. doi:10.1111/joim.13219.
- GreyNet International. Grey literature sources for systematic reviews: Reports, theses, and clinical trials registers. 2024. Available at: www.greynet.org
- OpenGrey. OpenGrey: A database for grey literature. 2024. Available at: www.opengrey.eu
- Cooke A, Smith D, Booth A. Beyond PICO: The SPIDER tool for qualitative evidence synthesis. Qual Health Res. 2012;22(10):1435-1443. doi:10.1177/1049732312452938.
- Alonso R, Muñiz-Grijalvo O, Díaz-Díaz JL, et al. Efficacy of PCSK9 inhibitors in the treatment of heterozygous familial hypercholesterolaemia: A clinical practice experience. J Clin Lipidol. 2021;15(4):584-592. doi:10.1016/j.jacl.2021.04.011.
- Duell PB, Gidding SS, Andersen RL, et al. Longitudinal low density lipoprotein cholesterol goal achievement and cardiovascular outcomes among adult patients with familial hypercholesterolaemia: The CASCADE FH registry. Atherosclerosis. 2019;289:85-93. doi:10.1016/j.atherosclerosis.2019.08.007.
- Humphries SE, Cooper JA, Capps N, et al. Coronary heart disease mortality in severe vs non-severe familial hypercholesterolaemia in the Simon Broome Register. Atherosclerosis. 2019;281:207-212. doi:10.1016/j.atherosclerosis.2018.11.014.
- Raal F, Durst R, Bi R, et al. Efficacy, safety, and tolerability of inclisiran in patients with homozygous familial hypercholesterolemia: Results from the ORION-5 randomised clinical trial. Circulation. 2024;149(5):354-362. doi:10.1161/CIRCULATIONAHA.122.063460.
- Toell T, Mayer L, Pechlaner R, et al. Familial hypercholesterolaemia in patients with ischaemic stroke or transient ischaemic attack. Eur J Neurol. 2018;25(2):260-267. doi:10.1111/ene.13485.
- Glynn L. A critical appraisal tool for library and information research. Libr Hi Tech 2006; 24(3): 387-399. Available at: www.researchgate.net/publication/235262791_A_critical_appraisal_tool_for_library_and_information_research
- Eldredge JD. Evidence-based librarianship: An overview. Bull Med Libr Assoc. 2000;88(4):289-302. Available at: https://pubmed.ncbi.nlm.nih.gov/11055296/
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021; 372: n71.
- Zamora A, Mata P, Alonso R, et al. Disparities in treatment and lipid goal attainment in women with familial hypercholesterolemia: A call for equity. Eur Heart J 2020; 6(4): 312-318.
- Zhang X. Outcomes of PCSK9 inhibitors: Does sex matter? JACC Adv. 2023;2(9):100667. doi:10.1016/j.jacadv.2023.100667.
- Balla S, Ekpo EP, Wilemon KA, et al. Women living with familial hypercholesterolaemia: Challenges and considerations surrounding their care. Curr Atheroscler Rep. 2020;22(10):60. doi:10.1007/s11883-020-00881-5.
- Paquette M, Faubert S, Saint-Pierre N, et al. Sex differences in LDL-C response to PCSK9 inhibitors: A real world experience. J Clin Lipidol. 2023;17(1):142-149. doi:10.1016/j.jacl.2022.12.002.
- Cholesterol Treatment Trialists’ (CTT) Collaboration, Fulcher J, O’Connell R, et al. Efficacy and safety of LDL-lowering therapy among men and women: Meta-analysis of individual data from 174,000 participants in 27 randomised trials. Lancet. 2015;385(9976):1397-1405. doi:10.1016/S0140-6736(14)61368-4.
- Schreuder MM, Hamkour S, Siegers KE, et al. LDL cholesterol targets rarely achieved in familial hypercholesterolaemia patients: A sex and gender-specific analysis. Atherosclerosis. 2023;384:117117. doi:10.1016/j.atherosclerosis.2023.03.022.
- European Society of Cardiology (ESC) and European Atherosclerosis Society (EAS). ESC/EAS Guideline for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Eur Heart J 2019; 41(1): 111–188. Available at: https://academic.oup.com/eurheartj/article/41/1/111/5556353
- Chella Krishnan K, Mehrabian M, Lusis AJ. Sex differences in metabolism and cardiometabolic disorders. Curr Opin Lipidol. 2018;29(5):404-410. doi:10.1097/MOL.0000000000000536.
- Goldberg AC, Banach M, Catapano AL, et al. Evaluation of the efficacy and safety of bempedoic acid in women and men: Pooled analyses from phase 3 trials. Atherosclerosis. 2023;384:117192. doi:10.1016/j.atherosclerosis.2023.117192.
- Bosco G, Di Giacomo Barbagallo F, Di Marco M, et al. The impact of SLCO1B1 rs4149056 on LDL-C target achievement after lipid-lowering therapy optimisation in men and women with familial hypercholesterolaemia. Front Endocrinol (Lausanne). 2024;15:1346152. doi:10.3389/fendo.2024.1346152.
- Bosco G, Di Giacomo Barbagallo F, Di Marco M, et al. Effect of inclisiran on lipid and mechanical vascular profiles in familial hypercholesterolaemia subjects: Results from a single lipid centre real-world experience. Prog Cardiovasc Dis. 2025;92:108-117. doi:10.1016/j.pcad.2025.05.008.
