Reference: March 2026 | Issue 3 | Vol 12 | Page 46
Heart failure impacts men and women differently, exhibiting distinct epidemiological patterns, clinical presentations, responses to treatment, and outcomes. This article, based on a presentation at the inaugural Women’s Heart Summit in January, 2026, at the Westbury Hotel in Dublin, explores these gender disparities. Although women have traditionally been viewed as underrepresented in heart failure trials, closer analysis shows that trial enrolment may more closely mirror disease prevalence.
Women are less likely to develop heart failure with reduced ejection fraction (HFrEF) due to lower rates of ischaemic heart disease and dilated cardiomyopathy, but are more prone to develop heart failure with preserved ejection fraction (HFpEF). Despite concerns regarding guideline relevance, women consistently show better survival rates than men, with approximately 20 per cent lower annual mortality.
Finally, this article examines sex-specific differences in pharmacological therapy, device therapy, and emerging treatments, while warning against over-interpreting subgroup analyses.
Introduction
When examining gender disparities in heart failure research, one quickly encounters statements suggesting that women are under-enrolled in clinical trials,1 are less likely to receive treatments such as implantable cardioverter-defibrillators (ICDs),2 and that current guidelines are based on studies that have enrolled predominantly middle-aged Caucasian men.3
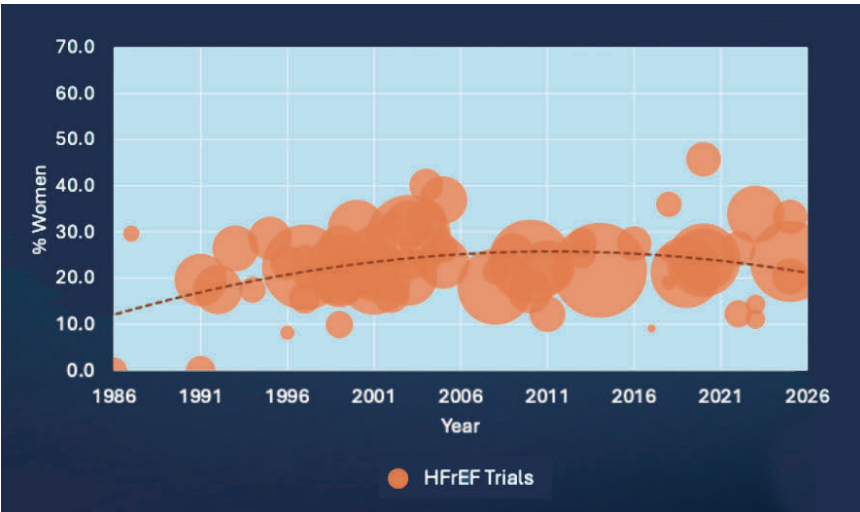
I looked at female participation in 66 landmark trials of medications and devices in patients with HFrEF, and women accounted for only 23.2 per cent of all participants (Figure 1). Superficially, the stereotypes appear accurate.
Furthermore, an analysis of authorship in key heart failure trials reveals a significant gender imbalance: Among the papers examined by Au et al,1 194 had male first authors, while only 30 had female first authors. Similarly, 193 men held senior author positions, compared to just 31 women.
This article will look at the changing demographic trends, including mortality and mode of death, and how treatments might affect men and women differently, before returning to look at female participation in heart failure trials and discussing some of the limitations of the literature.
Epidemiology of heart failure
Some experts have warned of a growing heart failure epidemic. However, data from the UK and the EU suggests that heart failure admissions are, if anything, falling slightly in the UK4 and by approximately 25-30 per cent across Europe.5 Data from Conrad et al showed an increase in the number of heart failure cases in the UK between 2002 and 2014, from just over 750,000 to almost 921,000.6
The latest data from the CVD Prevent database4 derived from general practice systems and combined with UK population estimates7 indicate there are currently around 907,600 cases (1.31% of 69,281,400) in the UK, suggesting little growth over the past decade.
Patient characteristics by ejection fraction phenotype
The typical characteristics of patients with HFrEF and HFpEF differ substantially. Patients with HFrEF are typically younger, predominantly male, have fewer comorbidities, and commonly present with ischaemic heart disease or dilated cardiomyopathy as the underlying aetiology. Ischaemic heart disease mortality data from the Office for National Statistics (2019) demonstrate that men outnumber women by 2:1.8
However, in later years, women begin to approach male rates without ever surpassing them. Epidemiological studies from the US and the UK examining dilated cardiomyopathy incidence similarly show men outnumbering women by just over 2:1.9,10
In contrast, patients with HFpEF are typically older, predominantly female, with women outnumbering men by approximately 2:1, frequently have multiple comorbidities, and commonly present with hypertension, valvular heart disease, atrial fibrillation, and obesity as contributory factors.3,8,11
Some studies have estimated that four in five patients with HFpEF are overweight or obese.12 The impact of obesity is probably more than just an ‘engine-chassis mismatch’,13 and a comprehensive analysis of the impact of obesity on the heart and the development of heart failure has recently been published by Milton Packer.14
Demographic trends
Data from the National Institute of Cardiovascular Outcomes Research (NICOR) shows that the underlying causes of heart failure are evolving. There has been a gradual decrease in patients with HFrEF and a corresponding increase in those with HFpEF.4 This trend has been observed across Europe and the USA.15
NICOR data indicate that at younger ages, men significantly outnumber women in heart failure cases, but as age increases, women catch up and eventually exceed men numerically by age 85. Overall, the latest data reveals that approximately 44 per cent of patients discharged from hospital with a diagnosis of heart failure are female, while 56 per cent are male.
The shift towards HFpEF and away from HFrEF will continue. The population is ageing: In the European Union, three in 10 people will be over 65 by 2050.5 Concurrently, the population is becoming increasingly obese.16
Outcomes in heart failure
Heart failure remains a lethal condition. In the UK, among patients discharged with a primary diagnosis of heart failure, about 10 per cent die during the initial hospital stay, and 12 per cent will have died within 30 days of discharge.4
Modern therapies have made a substantial difference. The contemporary ‘four pillars’ of heart failure therapy include angiotensin-converting enzyme (ACE) inhibitors, angiotensin II receptor antagonists (ARBs) or angiotensin receptor-neprilysin inhibitors (ARNIs), beta-blockers, mineralocorticoid receptor antagonists (MRAs), and sodium-glucose co-transporter 2 (SGLT2) inhibitors. This therapeutic approach has reduced two-year mortality from approximately 35 per cent to around 10 per cent.17
Importantly, when outcomes are examined by sex, women consistently fare better than men. A woman with heart failure is approximately 20 per cent less likely to die in any given year compared with a man.18,19 The reasons underlying this mortality difference are beyond the scope of this article.
Pharmacological treatment
This section explores sex-specific differences in the main pharmacological therapies.
ACE Inhibitors and ARBs: Many early ACE inhibitor trials did not report sex-specific outcomes.20,21,22 Those that did suggested no significant interaction with sex, with men and women deriving similar benefits.23,24 There have been suggestions that women might experience more adverse drug reactions, although these findings have not been consistent.25,26,27,28,29
Likewise, ARB trials showed no significant differences in effect between men and women.30,31,32
Santema et al33 made an interesting observation in a post hoc analysis of the BIOSTAT-CHF34 and ASIAN-HF35 studies. They noted that in men, the lowest risks of death and heart failure hospitalisation occurred at 100 per cent of guideline-recommended doses of ACE inhibitors and ARBs. In contrast, in women, the maximum benefit was achieved at 50 per cent of these doses.
ARNIs: Sex-specific responses to sacubitril/valsartan vary depending on ejection fraction phenotype. The PARADIGM-HF study36 examined patients with HFrEF and found no significant difference in the effects of sacubitril/valsartan between men and women. However, the PARAGON-HF37 study in patients with HFpEF suggested that women may gain considerably more benefit than men.
Beta-blockers: Many early beta-blocker trials did not report outcomes by sex in the main trial papers.38,39,40,41,42 Of those that did, most indicated no difference between men and women.43,44,45 Some studies suggested greater mortality reduction in men,46,47 while another found significant mortality reduction in women but not in men.48 Overall, there appears to be no clinically meaningful difference between the two groups.
However, there may be differences in the doses needed for men and women. Pharmacological studies have shown that plasma concentrations of beta-blockers can be up to 2.5 times higher at the same dose in women compared to men.49,50,51 The Santema study33 also indicated that women attain maximum benefit at just 50 per cent of the doses needed by men.
MRAs and SGLT2 inhibitors: For MRAs, no sex-specific differences in response have been observed in either HFrEF or HFpEF.52 The same seems to apply to SGLT2 inhibitors.53
Cardiac glycosides: Digoxin and digitoxin present more complex sex-specific considerations. The DIG study (1997)54 and the DIGIT-HF study (2025)55 both investigated patients with HFrEF. Interestingly, mortality rates were similar between studies despite the DIGIT-HF cohort being sicker, with a higher proportion of patients in NYHA Class III.
Post-hoc analysis of the DIG study by Rathore et al56 suggested a significant excess of deaths in women, but no adverse impact in men. However, it should be noted that digoxin doses in the DIG study were high (approximately 250 micrograms daily, with permitted doses up to 500 micrograms) – doses not commonly used today.
Analysis by Adams et al57 examining hazard ratios for mortality by serum digoxin concentration found little difference between men and women, but higher concentrations were associated with increased risk. Since women achieve higher digoxin levels for equivalent doses, this may explain the apparent sex difference.
In the DIGIT-HF study, no increase in mortality was observed, and the effect of digitoxin was similar in men and women. The critical difference was that serum digitoxin levels were monitored and doses adjusted to avoid toxic concentrations.
Glucagon-like peptide-1 receptor agonists: Analysis of the STEP-HFpEF58 and SUMMIT59 trials, which examined semaglutide and tirzepatide in patients with HfpEF and obesity, suggests that the benefits are consistent between women and men. However, women tend to achieve greater weight loss than men.
Device and other therapies
Implantable cardioverter-defibrillators (ICDs)
Women are less likely to die suddenly than men, which may affect the impact of ICDs on survival in women. A study by Rho et al examined differences in modes of death between men and women with heart failure who were eligible for primary prevention ICDs, analysing data from three trials and two registries. For any given Seattle Heart Failure Score, women had a 30 per cent lower risk of sudden cardiac death but a 54 per cent higher risk of dying from pump failure compared with men.
Overall, women had better survival than men.60
A similar pattern is seen in HFpEF. Dewan et al found that women generally have lower mortality rates compared with men, mainly due to a significantly reduced risk of sudden cardiac death.61 The causes become clearer when considering the reasons for sudden death; the most common cause is coronary artery disease, which, as described above, is much more common in men.
When ICDs are implanted, women have a much lower risk of first appropriate shock – approximately 1.1–1.2 per cent per year compared to 2.5–3 per cent per year for men.62 However, device therapy poses a greater risk for women. Russo et al demonstrated that women experience significantly higher rates of pneumothorax, haematoma, tamponade, and complications requiring system revision at the time of implant.63 Lead extraction risks are also considerably higher in women.64
Ghanbari et al conducted a meta-analysis of sex-specific outcomes in primary prevention ICD trials and found no significant mortality benefit for women, whereas men demonstrated a substantial reduction in mortality.65 It remains unclear whether women derive benefit from primary prevention ICDs, and further research is needed.66
Cardiac resynchronisation therapy (CRT)
The situation is reversed for CRT. Data from MADIT-CRT demonstrated a clear benefit of CRT-D over ICD in women, but no clear benefit in men.67 Furthermore, a meta-analysis of three CRT-D versus ICD trials (REVERSE, MADIT-CRT, and RAFT) showed substantially greater benefits for women than for men.68 This may be explained by physical size: A QRS duration of 150 milliseconds in a smaller heart is likely to cause greater desynchrony than the same QRS duration in a larger heart.69
Atrial fibrillation ablation
Atrial fibrillation frequently co-occurs with heart failure,70 including both HFpEF and HFrEF. Often, the dysfunction and symptoms are merely a result of atrial fibrillation, and ablation can be curative, restoring normal ventricular function.71,72,73 As a heart failure doctor, I treat heart failure. Electrophysiologists can cure heart failure. Atrial fibrillation ablation, which is arguably underused in the heart failure population, appears to be equally effective in both women and men with heart failure in clinical trials.74,75
Are women truly underrepresented?
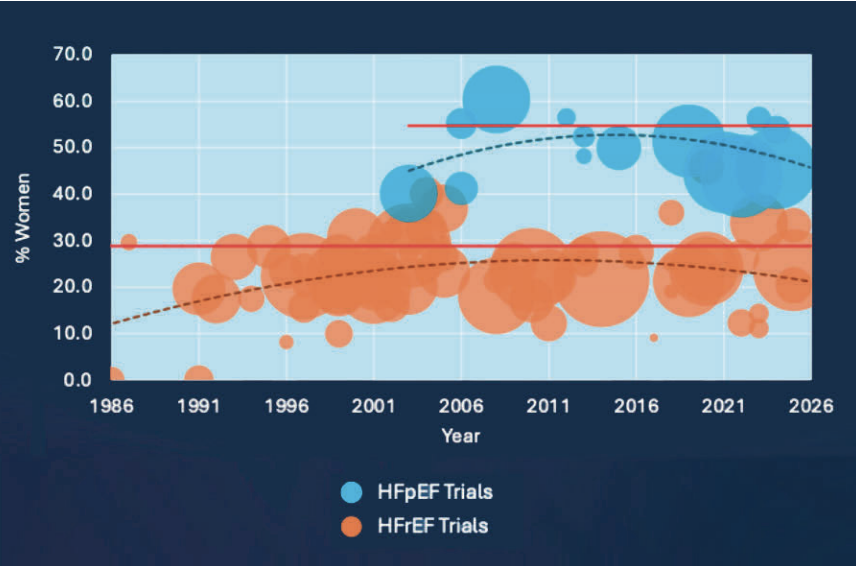
Although data suggest that women are under-represented in HFrEF trials, examination of HFpEF trials shows female participation closer to 50 per cent. Comparison with prevalence data from the SwedeHF registry suggests that the apparent under-representation of women in HFrEF trials may be because women less commonly present with this phenotype – the trials reflect the populations being studied (Figure 2). There is no systematic bias towards recruiting men.
The remaining gap is likely explained by trials’ tendency to recruit patients without comorbidities, and who are not of childbearing age, which makes studies more practical and facilitates more precise conclusions. This approach further skews enrolment toward male patients, since women only begin to outnumber men when they are very elderly. The disease pattern being studied is simply more common in men. However, some women may not choose to participate due to caring responsibilities, and there are data to suggest that when women lead trials, a higher proportion of women tend to be recruited.76
The limitations of subgroup analysis
Clinicians are accustomed to seeing extensive tables with multiple subgroup analyses, which can produce contradictory results. For example, in the VICTORIA trial (Vericiguat in patients with heart failure), patients with NYHA Class III or IV heart failure appeared to benefit from vericiguat. In contrast, patients with very high BNP (B-type natriuretic peptide) levels (which typically correlate with NYHA class) seemed to do worse.77 Such apparent contradictions reflect the random play of chance and the statistical consequences of multiple comparisons in progressively smaller subgroups.
Richard Peto vividly illustrated the folly of subgroup analysis in the ISIS-2 trial,78 which proved aspirin and streptokinase to be superior treatments for myocardial infarction. Reportedly compelled to discuss subgroup analysis, Peto maliciously complied, and tested outcomes by astrological sign. Patients born under Gemini and Libra performed slightly worse with aspirin, while those under all other signs gained notable benefit. The results can still be found in the manuscript published in The Lancet.
Similar cautionary tales exist within heart failure research – one does not have to look far. The PRAISE trial (1996) examined amlodipine in heart failure and, in a subgroup analysis, found that patients with dilated cardiomyopathy did substantially better with amlodipine than with placebo.79 This hypothesis was tested properly in PRAISE-2 (2013), which showed no difference in outcomes between amlodipine and placebo in dilated cardiomyopathy.80
The ELITE trial suggested that losartan was superior to captopril in heart failure, but the original trial was small and was not powered for mortality;81 the subsequent, much larger, ELITE-2 trial demonstrated no mortality difference between the two agents.30
If we really wish to understand actual differences in outcomes between men and women, trials must be specifically designed and powered to detect such differences. Until then, conclusions from subgroup analyses, and thus many of my conclusions above, must be interpreted with great caution.
Conclusions
Women have lower rates of ischaemic heart disease and dilated cardiomyopathy than men and are therefore less likely to develop HFrEF. Conversely, they are more likely to develop HFpEF. As the population ages and obesity rates increase, the number of women with HFpEF will continue to rise. At the same time, advances in the early treatment of ischaemic heart disease and improved detection and treatment of left ventricular systolic dysfunction should decrease the prevalence of HFrEF.
Although guidelines are mainly based on male-derived data, women are hospitalised with heart failure less often and have better survival rates. Women are more likely to die from pump failure and less likely to die suddenly.
Regarding pharmacological therapy, women may require lower doses of ACE inhibitors, ARBs, and beta-blockers. Women with HFpEF may gain greater benefit from sacubitril/valsartan. When using digoxin or digitoxin, careful monitoring of serum levels is essential, recognising that women reach higher concentrations than men for any given dose.
With device therapy, ICDs may be less beneficial for women, while CRT seems more advantageous. The benefit of ICDs in women still needs to be conclusively proven. Clinicians should be cautious with dosing and tailor medication doses to each patient rather than blindly follow guideline-target doses.
Although identifying sex-specific differences remains difficult, the growing use of electronic medical records could allow larger pragmatic trials to better understand these differences at lower cost.
While we must be cautious about over-interpreting subgroup analyses, the findings across multiple trials suggest genuine biological differences that warrant further investigation through appropriately designed and powered clinical trials.
Women are under-enrolled in clinical trials compared with men, but the differences are perhaps not as great as people think, and the populations enrolled reflect the sex distribution of the diseases under study.
Nonetheless, there is room for improvement. Understanding these issues exist, amending trial design, and increasing the number of women designing and leading trials should improve the situation, and help all patients with heart failure.
The full recording of the Women’s Heart Summit is available at:
Via MedCafe.ie: https://player.vimeo.com/video/1159395273?h=6a06aa7de0
On Mater Private website: https://www.materprivate.ie/for-healthcare-professionals/webinar-recordings
References
- Au M, Whitelaw S, Khan MS, et al. A systematic review of sex-specific reporting in heart failure clinical trials: Trial flow and results. JACC Adv. 2022;1(4):100079. doi:10.1016/j.jacadv.2022.100079.
- Hsich EM. Sex differences in advanced heart failure therapies. Circulation. 2019;139(8):1080–1093. doi:10.1161/CIRCULATIONAHA.118.037369.
- Ciutac AM, Pana T, Dawson D, Myint PK. Sex-related differences in heart failure patients: Physiological mechanisms of cardiovascular ageing and evidence-based sex-specific medical therapies. Ther Adv Cardiovasc Dis. 2025;19:17539447241309673. doi:10.1177/17539447241309673.
- National Institute for Cardiovascular Outcomes Research. National Heart Failure Audit 2024 Summary Report. 2025. Available at: www.nicor.org.uk/national-cardiac-audit-programme/heart-failure-audit-nhfa
- OECD. The State of Cardiovascular health in the European Union.
OECD Publishing; 2025. doi:10.1787/ea7a15f4-en. - Conrad N, Judge A, Tran J et al. Temporal trends and patterns in heart failure incidence: A population-based study of 4 million individuals. Lancet 2018;391:572–580.
- Office for National Statistics. UK Population estimates. Available at: www.ons.gov.uk/peoplepopulationandcommunity/populationandmigration/populationestimates
- Escobar C, Palacios B, Varela L, et al. Prevalence, characteristics, management, and outcomes of patients with heart failure with preserved, mildly reduced, and reduced ejection fraction in Spain. J Clin Med. 2022;11(17):5199. doi:10.3390/jcm11175199.
- Ababio Y, Kelly SP, Angeli FS, et al. Prevalence and clinical burden of idiopathic dilated cardiomyopathy in the US. Am J Med Open. 2023;10:100038. Published 2023 Feb 25. doi:10.1016/j.ajmo.2023.100038/.
- Myers MC, Berge A, Zhong Y, et al. Prevalence and incidence of dilated cardiomyopathy in the US and Western Europe: A systematic review. Cardiol Res. 2025;16(4):295–305. Published 2025 Jul 8. doi:10.14740/cr2071.
- Lee DS, Gona P, Vasan RS, et al. Relation of disease pathogenesis and risk factors to heart failure with preserved or reduced ejection fraction: Insights from the Framingham heart study of the national heart, lung, and blood institute. Circulation. 2009;119(24):3070-3077. doi:10.1161/CIRCULATIONAHA.108.815944.
- Kitzman DW, Lam CSP. Obese heart failure with preserved ejection fraction phenotype: From pariah to central player. Circulation. 2017;136(1):20-23. doi:10.1161/CIRCULATIONAHA.117.028365.
- Foulkes SJ, Moura-Ferreira S, Milani M, et al. Mechanisms of exercise limitation in heart failure with preserved ejection fraction and obesity: A case of engine-chassis mismatch. Am J Physiol Heart Circ Physiol. 2025;329(5):H1126-H1136. doi:10.1152/ajpheart.00587.2025.
- Packer M. The adipokine hypothesis of heart failure with a preserved ejection fraction: A novel framework to explain pathogenesis and guide treatment. J Am Coll Cardiol. 2025;86(16):1269–1373. doi:10.1016/j.jacc.2025.06.055.
- Shahim B, Kapelios CJ, Savarese G, Lund LH. Global public health burden of heart failure: An updated review. Card Fail Rev. 2023;9:e11. Published 2023 Jul 27. doi:10.15420/cfr.2023.05.
- NHS England. Health Survey for England 2024: Data Tables. Published 2024. Available at: https://digital.nhs.uk/data-and-information/publications/statistical/health-survey-for-england/2024/health-survey-for-england-2024-data-tables
- Bassi NS, Ziaeian B, Yancy CW, Fonarow GC. Association of optimal implementation of sodium-glucose cotransporter 2 inhibitor therapy with outcome for patients with heart failure. JAMA Cardiol. 2020;5(8):948–951. doi:10.1001/jamacardio.2020.0898.
- Qiu W, Wang W, Wu S, et al. Sex differences in long-term heart failure prognosis: A comprehensive meta-analysis. Eur J Prev Cardiol. 2024;31(17):2013–2023. doi:10.1093/eurjpc/zwae256.
- Walli-Attaei M, Joseph P, Johansson I, et al. Characteristics, management, and outcomes in women and men with congestive heart failure in 40 countries at different economic levels: An analysis from the Global Congestive Heart Failure (G-CHF) registry. Lancet Glob Health. 2024;12(3):e396–e405. doi:10.1016/S2214-109X(23)00557-0.
- CONSENSUS Trial Study Group. Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). N Engl J Med. 1987;316(23):1429–1435. doi:10.1056/NEJM198706043162301.
- SOLVD Investigators, Yusuf S, Pitt B, Davis CE, Hood WB, Cohn JN. Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. N Engl J Med. 1991;325(5):293–302. doi:10.1056/NEJM199108013250501.
- Packer M, Poole-Wilson PA, Armstrong PW, et al. Comparative effects of low and high doses of the angiotensin-converting enzyme inhibitor, lisinopril, on morbidity and mortality in chronic heart failure. ATLAS Study Group. Circulation. 1999;100(23):2312–2318. doi:10.1161/01.cir.100.23.2312.
- Effect of ramipril on mortality and morbidity of survivors of acute myocardial infarction with clinical evidence of heart failure. The Acute Infarction Ramipril Efficacy (AIRE) Study Investigators. Lancet. 1993;342(8875):821–828.
- Swedberg K, Held P, Kjekshus J, et al. Effects of the early administration of enalapril on mortality in patients with acute myocardial infarction. Results of the Cooperative New Scandinavian Enalapril Survival Study II (CONSENSUS II). N Engl J Med. 1992;327(10):678–684. doi:10.1056/NEJM199209033271002.
- Kostis JB, Shelton B, Gosselin G, et al. Adverse effects of enalapril in the studies of left ventricular dysfunction (SOLVD). SOLVD investigators. Am Heart J. 1996;131(2):350–355. doi:10.1016/s0002-8703(96)90365-8.
- Kostis JB, Kim HJ, Rusnak J, et al. Incidence and characteristics of angioedema associated with enalapril. Arch Intern Med. 2005;165(14):1637–1642. doi:10.1001/archinte.165.14.1637.
- Ishani A, Weinhandl E, Zhao Z, et al. Angiotensin-converting enzyme inhibitor as a risk factor for the development of anaemia, and the impact of incident anaemia on mortality in patients with left ventricular dysfunction. J Am Coll Cardiol. 2005;45(3):391–399. doi:10.1016/j.jacc.2004.10.038.
- Sadanaga T, Yoshimura M, Sakamoto T, Sumida H, Ogawa H. Enalapril-induced cough is associated with non-severe heart failure. Int J Cardiol. 2009;135(2):275-276. doi:10.1016/j.ijcard.2008.03.063.
- Do TP, Seetasith A, Belleli R, et al. A Database cohort study to assess the risk of angioedema among patients with heart failure initiating angiotensin-converting enzyme inhibitors in the US. Am J Cardiovasc Drugs. 2018;18(3):205–211. doi:10.1007/s40256-017-0256-x.
- Pitt B, Poole-Wilson PA, Segal R, et al. Effect of losartan compared with captopril on mortality in patients with symptomatic heart failure: Randomised trial – the Losartan Heart Failure Survival Study ELITE II. Lancet. 2000;355(9215):1582–1587. doi:10.1016/s0140-6736(00)02213-3.
- Cohn JN, Tognoni G; Valsartan Heart Failure Trial Investigators. A randomised trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N Engl J Med. 2001;345(23):1667–1675. doi:10.1056/NEJMoa010713.
- Granger CB, McMurray JJ, Yusuf S, et al. Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function intolerant to angiotensin-converting-enzyme inhibitors: the CHARM-Alternative trial. Lancet. 2003;362(9386):772–776. doi:10.1016/S0140-6736(03)14284-5.
- Santema BT, Ouwerkerk W, Tromp J et al. Identifying optimal doses of heart failure medications in men compared with women: A prospective, observational, cohort study. Lancet 2019;394:1254–1263.
- Ouwerkerk W, Voors AA, Anker SD, et al. Determinants and clinical outcome of uptitration of ACE-inhibitors and beta-blockers in patients with heart failure: A prospective European study. Eur Heart J. 2017;38(24):1883–1890. doi:10.1093/eurheartj/ehx026.
- Lam CS, Anand I, Zhang S, et al. Asian sudden cardiac death in heart failure (ASIAN-HF) registry. Eur J Heart Fail. 2013;15(8):928–936. doi:10.1093/eurjhf/hft045.
- McMurray JJ, Packer M, Desai AS, et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med. 2014;371(11):993–1004. doi:10.1056/NEJMoa1409077.
- Solomon SD, McMurray JJV, Anand IS, et al. Angiotensin-neprilysin inhibition in heart failure with preserved ejection fraction. N Engl J Med. 2019;381(17):1609–1620. doi:10.1056/NEJMoa1908655.
- Waagstein F, Bristow MR, Swedberg K, et al. Beneficial effects of metoprolol in idiopathic dilated cardiomyopathy. Metoprolol in Dilated Cardiomyopathy (MDC) Trial Study Group. Lancet. 1993;342(8885):1441–1446. doi:10.1016/0140-6736(93)92930-r.
- A randomised trial of beta-blockade in heart failure. The Cardiac Insufficiency Bisoprolol Study (CIBIS). CIBIS investigators and committees. Circulation. 1994;90(4):1765–1773. doi:10.1161/01.cir.90.4.1765.
- Randomised, placebo-controlled trial of carvedilol in patients with congestive heart failure due to ischaemic heart disease. Australia/New Zealand Heart Failure Research Collaborative Group. Lancet. 1997;349(9049):375–380.
- The Cardiac Insufficiency Bisoprolol Study II (CIBIS-II): A randomised trial. Lancet. 1999;353(9146):9–13. Available at: https://pubmed.ncbi.nlm.nih.gov/10023943/
- Dargie HJ. Effect of carvedilol on outcome after myocardial infarction in patients with left-ventricular dysfunction: The CAPRICORN randomised trial. Lancet. 2001;357(9266):1385–1390. doi:10.1016/s0140-6736(00)04560-8.
- Packer M, Bristow MR, Cohn JN, et al. The effect of carvedilol on morbidity and mortality in patients with chronic heart failure. US Carvedilol Heart Failure Study Group. N Engl J Med. 1996;334(21):1349–1355. doi:10.1056/NEJM199605233342101.
- Beta-Blocker Evaluation of Survival Trial Investigators, Eichhorn EJ, Domanski MJ, Krause-Steinrauf H, Bristow MR, Lavori PW. A trial of the beta-blocker bucindolol in patients with advanced chronic heart failure. N Engl J Med. 2001;344(22):1659–1667. doi:10.1056/NEJM200105313442202.
- Packer M, Coats AJ, Fowler MB, et al. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med. 2001;344(22):1651-1658. doi:10.1056/NEJM200105313442201.
- Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet. 1999;353(9169):2001–2007.
- Poole-Wilson PA, Swedberg K, Cleland JG, et al. Comparison of carvedilol and metoprolol on clinical outcomes in patients with chronic heart failure in the Carvedilol Or Metoprolol European Trial (COMET): Randomised controlled trial. Lancet. 2003;362(9377):7–13. doi:10.1016/S0140-6736(03)13800-7.
- Flather MD, Shibata MC, Coats AJ, et al. Randomised trial to determine the effect of nebivolol on mortality and cardiovascular hospital admission in elderly patients with heart failure (SENIORS). Eur Heart J. 2005;26(3):215–225. doi:10.1093/eurheartj/ehi115.
- Eugene AR. Metoprolol dose equivalence in adult men and women based on gender differences: Pharmacokinetic modelling and simulations. Med Sci (Basel). 2016;4(4):18. doi:10.3390/medsci4040018.
- Eugene AR. Gender based dosing of metoprolol in the elderly using population pharmacokinetic modeling and simulations. Int J Clin Pharmacol Toxicol. 2016;5(3):209–215.
- Soldin OP, Mattison DR. Sex differences in pharmacokinetics and pharmacodynamics. Clin Pharmacokinet. 2009;48(3):143–157. doi:10.2165/00003088-200948030-00001.
- Rossello X, Ferreira JP, Pocock SJ, et al. Sex differences in mineralocorticoid receptor antagonist trials: A pooled analysis of three large clinical trials. Eur J Heart Fail. 2020;22(5):834–844. doi:10.1002/ejhf.1740.
- Shah AJ, Vorla M, Jafri SH, Kalra D. SGLT2 inhibitors in women and cardiovascular outcomes – meta-analysis of sex differences in 11 randomised controlled clinical trials. J Am Coll Cardiol. 2024;83(4):321. doi:10.1016/j.cardfail.2023.10.320.
- Digitalis Investigation Group. The effect of digoxin on mortality and morbidity in patients with heart failure. N Engl J Med. 1997;336(8):525–533. doi:10.1056/NEJM199702203360801.
- Bavendiek U, Großhennig A, Schwab J, et al. Digitoxin in patients with heart failure and reduced ejection fraction. N Engl J Med. 2025;393(12):1155–1165. doi:10.1056/NEJMoa2415471.
- Rathore SS, Wang Y, Krumholz HM. Sex-based differences in the effect of digoxin for the treatment of heart failure. N Engl J Med. 2002;347(18):1403–1411. doi:10.1056/NEJMoa021266.
- Adams KF Jr, Patterson JH, Gattis WA, et al. Relationship of serum digoxin concentration to mortality and morbidity in women in the digitalis investigation group trial: A retrospective analysis. J Am Coll Cardiol. 2005;46(3):497–504. doi:10.1016/j.jacc.2005.02.091.
- Kosiborod MN, Abildstrøm SZ, Borlaug BA, et al. Semaglutide in patients with heart failure with preserved ejection fraction and obesity. N Engl J Med. 2023;389(12):1069–1084. doi:10.1056/NEJMoa2306963.
- Packer M, Zile MR, Kramer CM, et al. Tirzepatide for heart failure with preserved ejection fraction and obesity. N Engl J Med. 2025;392(5):427–437. doi:10.1056/NEJMoa2410027.
- Rho RW, Patton KK, Poole JE, et al. Important differences in mode of death between men and women with heart failure who would qualify for a primary prevention implantable cardioverter-defibrillator. Circulation. 2012;126(20):2402–2407. doi:10.1161/CIRCULATIONAHA.111.069245.
- Dewan P, Rørth R, Raparelli V, et al. Sex-related differences in heart failure with preserved ejection fraction. Circ Heart Fail. 2019;12(12):e006539. doi:10.1161/CIRCHEARTFAILURE.119.006539.
- Sticherling C, Arendacka B, Svendsen JH, et al. Sex differences in outcomes of primary prevention implantable cardioverter-defibrillator therapy: Combined registry data from eleven European countries. Europace. 2018;20(6):963–970. doi:10.1093/europace/eux176.
- Russo AM, Daugherty SL, Masoudi FA, Wang Y, Curtis J, Lampert R. Gender and outcomes after primary prevention implantable cardioverter-defibrillator implantation: Findings from the National Cardiovascular Data Registry (NCDR). Am Heart J. 2015;170(2):330–338. doi:10.1016/j.ahj.2015.02.025.
- Borkowski P, Schiavone M, Zou FJ, et al. Sex differences in periprocedural safety outcomes and mortality following cardiac implantable electronic device transvenous lead extraction. J Cardiovasc Electrophysiol. 2026;37(1):17–25. doi:10.1111/jce.70170.
- Ghanbari H, Dalloul G, Hasan R, et al. Effectiveness of implantable cardioverter-defibrillators for the primary prevention of sudden cardiac death in women with advanced heart failure: A meta-analysis of randomised controlled trials. Arch Intern Med. 2009;169(16):1500–1506. doi:10.1001/archinternmed.2009.255.
- Ghanbari H, Machado C. ICD therapy in women: Are men from Mars and women from Venus? Heart Rhythm. 2010;7(7):883–884. doi:10.1016/j.hrthm.2010.04.024.
- Arshad A, Moss AJ, Foster E, et al. Cardiac resynchronisation therapy is more effective in women than in men: The MADIT-CRT (Multicenter Automatic Defibrillator Implantation Trial with Cardiac Resynchronisation Therapy) trial. J Am Coll Cardiol. 2011;57(7):813–820. doi:10.1016/j.jacc.2010.06.061.
- Zusterzeel R, Selzman KA, Sanders WE, et al. Cardiac resynchronisation therapy in women: US Food and Drug Administration meta-analysis of patient-level data. JAMA Intern Med. 2014;174(8):1340–1348. doi:10.1001/jamainternmed.2014.2717.
- Zusterzeel R, Selzman KA, Sanders WE, et al. Toward sex-specific guidelines for cardiac resynchronisation therapy? J Cardiovasc Transl Res. 2016;9(1):12–22. doi:10.1007/s12265-015-9663-z.
- Yoshimura H, Paliwal N, Gonzalez-Izquierdo A, et al. Atrial fibrillation development in the heart failure population from nationwide British linked electronic health records. ESC Heart Fail. 2025;12(4):2507–2517. doi:10.1002/ehf2.15264.
- Segan L, Kistler PM, Nanayakkara S, et al. Withdrawal of heart failure therapy after atrial fibrillation rhythm control with ejection fraction normalisation: The WITHDRAW-AF trial. Eur Heart J. 2026;47(2):250–262. doi:10.1093/eurheartj/ehaf563.
- Al-Aidarous S, Rajappan S, Ahluwalia N, et al. Left ventricular ejection fraction response after atrial fibrillation ablation: Clinical predictors and development of the PACED score. Heart Rhythm. 2026;23(3):e392–e402. doi:10.1016/j.hrthm.2025.11.039.
- Al-Aidarous S, Rajappan S, Ahluwalia N, et al. Withdrawing guideline-directed medical therapy after left ventricular ejection fraction recovery following atrial fibrillation ablation: A multicentre cohort study. Open Heart. 2025;12(2):e003733. doi:10.1136/openhrt-2025-003733.
- Fink T, Moersdorf M, Costard-Jaeckle A, et al. Sex and treatment outcomes of atrial fibrillation ablation in end-stage heart failure: Insights from CASTLE-HTx. JACC Heart Fail. 2025;13(10):102608. doi:10.1016/j.jchf.2025.102608.
- Marrouche NF, Brachmann J, Andresen D, et al. Catheter ablation for atrial fibrillation with heart failure. N Engl J Med. 2018;378(5):417–427. doi:10.1056/NEJMoa1707855.
- Chhaya VY, Binion CC, Mulles SM, et al. Gender bias in clinical trial rnrollment: Female authorship matters. Ann Vasc Surg. 2023;95:233–243. doi:10.1016/j.avsg.2023.03.008.
- Armstrong PW, Pieske B, Anstrom KJ, et al. Vericiguat in patients with heart failure and reduced ejection fraction. N Engl J Med. 2020;382(20):1883–1893. doi:10.1056/NEJMoa1915928.
- Randomised trial of intravenous streptokinase, oral aspirin, both, or neither among 17,187 cases of suspected acute myocardial infarction: ISIS-2.ISIS-2 (Second International Study of Infarct Survival) Collaborative Group. J Am Coll Cardiol. 1988;12(6 Suppl A):3A-13A. doi:10.1016/0735-1097(88)92635-6.
- Packer M, O’Connor CM, Ghali JK, et al. Effect of amlodipine on morbidity and mortality in severe chronic heart failure. Prospective Randomised Amlodipine Survival Evaluation Study Group. N Engl J Med. 1996;335(15):1107–1114. doi:10.1056/NEJM199610103351504.
- Packer M, Carson P, Elkayam U, et al. Effect of amlodipine on the survival of patients with severe chronic heart failure due to a nonischemic cardiomyopathy: Results of the PRAISE-2 study (prospective randomised amlodipine survival evaluation 2). JACC Heart Fail. 2013;1(4):308–314. doi:10.1016/j.jchf.2013.04.004.
- Pitt B, Segal R, Martinez FA, et al. Randomised trial of losartan versus captopril in patients over 65 with heart failure (Evaluation of Losartan in the Elderly Study, ELITE). Lancet. 1997;349(9054):747–752. doi:10.1016/s0140-6736(97)01187-2.
