Reference: March 2026 | Issue 3 | Vol 12 | Page 56
Cardiac implantable electronic devices (CIEDs) have transformed the management of bradyarrhythmias, malignant ventricular arrhythmias, and selected forms of heart failure. Their origins can be traced to 1958 in Stockholm, when a 42-year-old Swedish patient, Arne Larsson, underwent implantation of the first fully implantable pacemaker for recurrent Stokes-Adams attacks due to complete atrioventricular (AV) block.
At the time, management of severe bradyarrhythmias was limited to bed rest, short-acting pharmacological therapy, and externally powered pacing systems that were temporary, unreliable, and associated with significant morbidity. As such, syncope-related injuries, progression to heart failure, and sudden cardiac death were common.
The innovation introduced by surgeon Åke Senning and physician Rune Elmqvist consisted of a simple pulse generator implanted subcutaneously and connected to epicardial leads. Although the first device functioned only a few hours, the concept itself proved durable, and marked the transition from externally powered rescue therapy to continuous internal rhythm support. Arne went on to receive over 25 devices in his lifetime, outliving both his pioneering surgeon and physician.
Subsequent decades saw the development of implantable cardioverter-defibrillators (ICDs), cardiac resynchronisation therapy (CRT) systems, and implantable cardiac monitors, expanding the role of CIEDs beyond bradyarrhythmia management to include sudden cardiac death prevention, heart failure therapy, and long-term rhythm surveillance.
Globally, millions of people now live with a CIED.1 In Ireland, the National Cardiac Device Implant Registry has recorded more than 100,000 cardiac device implants to date.2 Rising implantation rates and improved survival mean that CIED recipients are increasingly encountered across primary care, acute medicine, and peri-operative practice.
For the general clinician, familiarity with device indications, follow-up principles, and recent technological developments is therefore essential. This review outlines contemporary CIED therapy types, indications, potential complications, and highlights recent updates relevant to everyday clinical care.
Overview of CIED types
PERMANENT PACEMAKERS
Permanent pacing is the definitive therapy for symptomatic bradycardia due to intrinsic conduction disease, most commonly sinus node dysfunction and high-grade AV block (Table 1). Both conditions largely reflect age-related degeneration of the cardiac conduction system, and pacemaker implantation is therefore predominantly a therapy of older adults, with more than 80 per cent of devices implanted in patients over 65 years.
Asymptomatic bradycardia, such as that seen in athletes with high vagal tone, does not require treatment. Nocturnal bradycardia alone is not an indication for pacing, and reversible causes such as drug toxicity, metabolic disturbance or electrolyte imbalance should be excluded before device implantation.3
Indications: Sinus node dysfunction
Sinus node dysfunction (sick sinus syndrome) encompasses disorders of sinoatrial node function including sinus bradycardia, sinoatrial block, sinus arrest, chronotropic incompetence, and bradycardia-tachycardia syndrome. Symptoms typically include fatigue, presyncope, syncope, and exercise intolerance.
The tachycardia-bradycardia variant is most common and reflects fibrosis of the sinus node and surrounding atrial myocardium. Episodes of atrial tachyarrhythmia, usually atrial fibrillation, alternate with sinus pauses due to overdrive suppression of the diseased sinus node, which is unable to promptly resume normal pacemaker activity at arrhythmia termination.
Pharmacological rate control of the tachyarrhythmia, such as beta-blockers, may worsen the periods of bradycardia, and therefore, pacemaker implantation can permit safe administration of rate-controlling medications. Unlike high-grade AV block, pacemaker implantation in sinus node dysfunction has not been shown to confer a mortality benefit, and pacing is therefore recommended only when symptoms are clearly attributable to bradycardia.3
| CLINICAL CATEGORY |
PACEMAKER RECOMMENDED |
PACEMAKER MAY BE CONSIDERED |
PACEMAKER NOT INDICATED |
|---|---|---|---|
| Sinus node dysfunction |
Symptomatic sinus bradycardia, sinus arrest, or sinoatrial block |
Asymptomatic sinus bradycardia | |
| Atrioventricular block | Third-degree AV block; second- degree Mobitz II or high-grade AV block |
Symptomatic first- degree AV block with marked PR prolongation |
First-degree AV block without symptoms; asymptomatic Wenckebach |
| Syncope with conduction disease |
Syncope with documented pause-related bradycardia or AV block |
Unexplained syncope with bifascicular block and evidence of conduction disease |
Syncope without evidence of bradyarrhythmia |
| Atrial fibrillation with bradycardia |
Symptomatic slow ventricular response or prolonged pauses |
Asymptomatic slow ventricular rate | |
| Postprocedure conduction disease |
Persistent high-grade AV block after cardiac surgery or transcatheter valve implantation |
Transient AV block with high-risk of recurrence |
Transient AV block that resolves |
| Reversible causes | Bradycardia due to medications, electrolyte disturbance, or acute illness |
||
| General considerations |
Limited life expectancy (<1 year) or severe competing comorbidity |
TABLE 1: Indications for permanent pacemaker implantation, stratified by clinical scenario and recommendation strength
AV = Atrioventricular; AF = Atrial fibrillation.
Adapted from: 2021 ESC Guidelines on cardiac pacing and cardiac resynchronisation therapy
Indications: AV Block
Management of AV block, meanwhile, aims to relieve symptoms and prevent progression to heart failure or sudden cardiac death. First-degree and Mobitz I (Wenckebach) AV block rarely progress and usually do not require pacing in asymptomatic patients.
In contrast, Mobitz II, or third-degree AV block, carries a high risk of syncope and progression to complete block, and pacing is generally recommended once reversible causes are excluded.3
RATE-RESPONSE THERAPY
Chronotropic incompetence describes failure to appropriately increase heart rate with metabolic demand. Although recognised as a subtype of sinus node dysfunction, it is not an independent indication for pacing and more commonly occurs in broader conduction disease, heart failure, or following an ablate-and-pace strategy.3,4
Rate-responsive pacing attempts to reproduce physiological heart-rate increases during exertion using accelerometers or minute-ventilation sensors. While this may modestly improve exercise capacity, consistent improvements in quality of life are less clear,5 and benefits in heart failure populations appear limited.6
Inappropriate programming may cause symptoms, with excessive rate augmentation producing palpitations (eg, when passive motion while on a vehicle is interpreted as activity), whereas inadequate augmentation may result in persistent exertional intolerance. Patients presenting with such symptoms should therefore be referred for device reprogramming.3
TYPES OF PACING SYSTEM – TRANSVENOUS
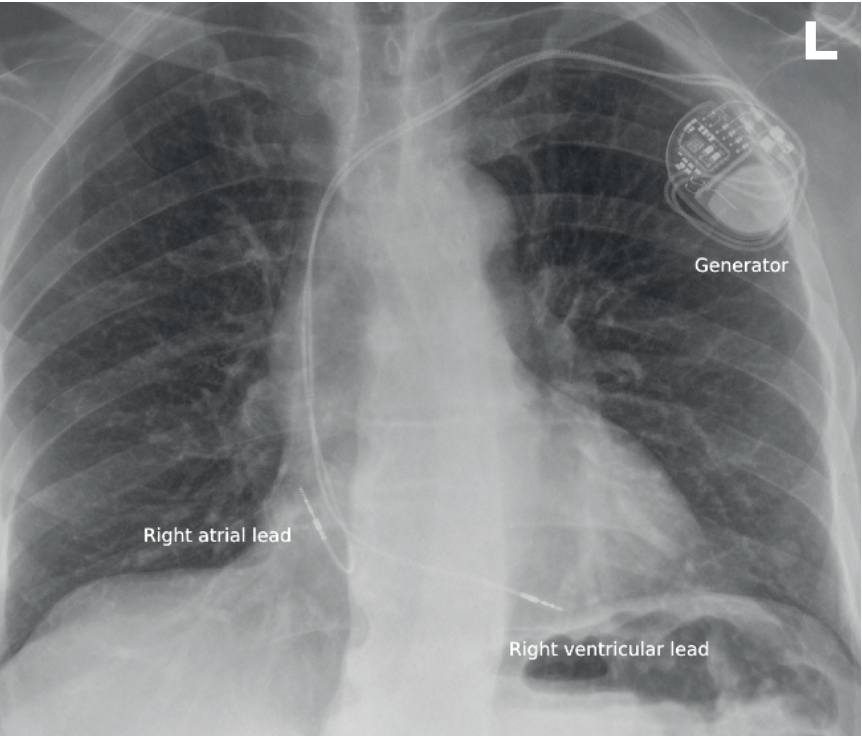
Most pacemakers are transvenous systems comprising a titanium pulse generator implanted in a subcutaneous chest wall pocket and connected to the myocardium via insulated leads introduced through the venous system (Figure 1).
Leads are typically secured to the endocardium using active screw fixation. Devices are usually implanted on the left side due to more straightforward venous access and avoidance of the dominant arm, although right-sided placement may be required in patients with venous access issues or prior infection.
Contemporary pacemakers use bipolar leads with closely spaced tip and ring electrodes, confining the pacing circuit to a small region of myocardium. This requires only a limited tissue volume for sensing and capture, creating pacing artefacts (‘spikes’) that are often too small to be readily visible on surface electrocardiogram (ECG).
Hence, one of several reasons why the absence of a visible pacing spike does not imply device malfunction, and the surface ECG alone cannot reliably distinguish between a functioning and a non-functioning pacemaker.
PACEMAKER CLASSIFICATION
Pacemaker systems are traditionally classified according to lead number and location. Dual-chamber devices with right atrial and right ventricular leads account for approximately two-thirds of implants, while the remainder are predominantly single-chamber ventricular systems.7
Devices are described using a standardised three- to five-letter code indicating which chambers are paced and sensed and the device response to intrinsic activity. For example, VVI denotes ventricular pacing and sensing with inhibition when intrinsic activity is detected, while DDD denotes dual-chamber pacing and sensing with coordinated atrioventricular timing (Table 2). Additional letters indicate features such as rate-responsive pacing (R).
Dual-chamber systems are commonly used in sinus node dysfunction and AV block in patients in sinus rhythm. Historically, atrial-only pacing was used in selected sinus node disease, where AV conduction was intact. However, given that approximately 10 per cent of patients later develop AV conduction disease requiring ventricular pacing,⁸ dual-chamber devices are now generally preferred.
Single-chamber ventricular pacing may not be appropriate for younger patients without atrial fibrillation, as loss of AV synchrony allows for atrial contraction against a closed mitral valve, leading to a ‘pacemaker syndrome’, characterised by fatigue, breathlessness, hypotension, and cannon A waves.
It is therefore primarily used in patients with permanent atrial fibrillation and slow ventricular response. Alternatively, in patients with refractory rapid atrial fibrillation, AV node ablation may be performed, rendering them pacemaker dependent while permitting reliable control of ventricular rate, the so-called ‘ablate and pace strategy’.
| POSITION | MEANING | OPTIONS |
|---|---|---|
| 1st | Chamber paced | O = none, A = atrium, V = ventricle, D = dual |
| 2nd | Chamber sensed | O, A, V, D |
| 3rd | Response to sensing | O = none, I = inhibited, T = triggered, D = dual (triggered + inhibited) |
| 4th | Rate modulation | O = none, R = rate responsive |
| 5th | Multisite pacing | O = none, A = atrial, V = ventricular, D = dual |
TABLE 2: Standard pacemaker mode nomenclature (NBG code) describing chamber pacing, chamber sensing, and response to sensing, with optional rate modulation and multisite pacing designations.
Adapted from: 2021 ESC Guidelines on cardiac pacing and cardiac resynchronisation therapy
PACEMAKER-INDUCED CARDIOMYOPATHY
Right ventricular apical pacing activates the ventricle outside the His-Purkinje system, producing slow myocardial conduction and electrical dyssynchrony resembling left bundle branch block. In susceptible individuals, this may lead to adverse ventricular remodelling, progressive systolic dysfunction, and increased atrial fibrillation risk,8 a condition termed ‘pacemaker-induced cardiomyopathy’.
Risk increases with greater right ventricular pacing burden, particularly when pacing exceeds 20-40 per cent over one to two years.8 Patients with impaired left ventricular function or baseline QRS prolongation are particularly vulnerable.
Devices are therefore programmed to promote intrinsic AV conduction where safely possible, with pacing burden reviewed during follow-up. Higher-risk patients should undergo periodic echocardiographic surveillance, with reprogramming or upgrade to physiologic pacing considered if ventricular dysfunction develops.3
CARDIAC PHYSIOLOGICAL PACING- PACING – CRT
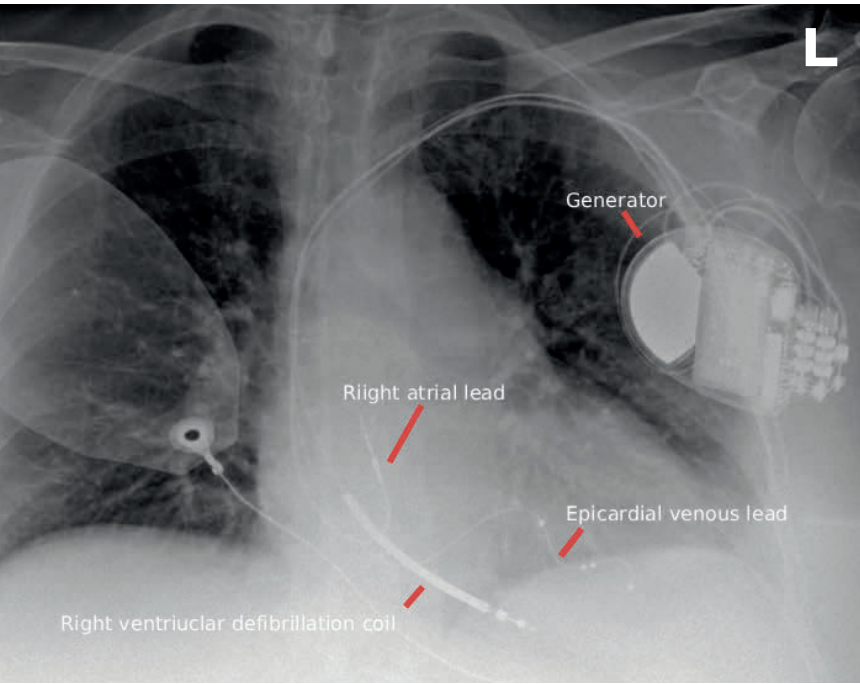
In contrast to conventional right ventricular apical pacing, cardiac physiologic pacing aims to preserve or restore ventricular synchrony. The first widely adopted approach was CRT. Emerging in the late 1990s and gaining broad acceptance in the early 2000s, CRT added a left ventricular lead to a conventional right atrial and right ventricular system. This lead is advanced via the coronary sinus and positioned in a lateral or posterolateral epicardial vein, enabling coordinated biventricular stimulation (Figure 2).
The concept was initially mechanical – if heart failure involved delayed contraction of opposing ventricular walls, pacing both ventricles together might restore coordinated pump function. This was supported by recognition that QRS prolongation and electromechanical dyssynchrony, particularly in left bundle branch block, contribute to systolic dysfunction.8
Early haemodynamic studies showed acute improvements in stroke volume, cardiac output, and mitral regurgitation.9 Subsequent randomised trials demonstrated improvements in symptoms, reverse remodelling, reduced heart failure hospitalisation, and reduced mortality.
Multiple landmark randomised control trials9,10,11 established CRT as disease-modifying therapy in patients with symptomatic heart failure, left ventricular ejection fraction (LVEF) ≤35 per cent, sinus rhythm, left bundle branch block, and QRS ≥150ms despite guideline-directed medical therapy. Emerging data suggest some subgroups, including women, may benefit at QRS durations of 120-149ms.8
CRT systems are more costly and complex than standard dual-chamber devices, and left ventricular lead implantation may be technically challenging due to variable coronary venous anatomy, with risks including lead instability, phrenic nerve stimulation, and greater procedural complexity.
PHYSIOLOGICAL PACING: CONDUCTION SYSTEM PACING
If CRT represented a mechanical solution to dyssynchrony, conduction system pacing aims to recruit the native His-Purkinje network directly. Early approaches focused on right ventricular septal pacing, but have evolved to direct His bundle pacing and, more recently, left bundle branch area pacing – restoring near-normal ventricular activation through the heart’s specialised conduction tissue.
Although the evidence base has not yet matured to that of CRT, observational studies and emerging randomised data support its use in patients requiring substantial ventricular pacing, particularly those at risk of pacing-induced cardiomyopathy, such as patients undergoing an ablate-and-pace strategy for refractory atrial fibrillation.
The ability to physiologically pace the heart has begun to change the risk/benefit ratio for such patients, in whom the risks of pacemaker induced cardiomyopathy may have outweighed potential benefits. Current guidelines endorse conduction system pacing or CRT in patients with reduced left ventricular function or anticipated high ventricular pacing burden, including those undergoing AV node ablation.
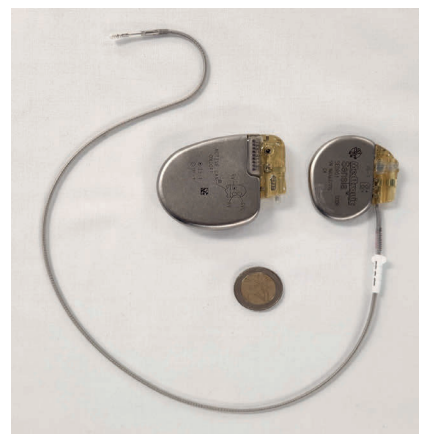
LEADLESS PACEMAKERS
Leadless pacemakers represent another innovation in pacing. These miniaturised devices (~2.5cm long, <2g) are implanted entirely within the right ventricle and avoid transvenous leads and subcutaneous pockets, reducing infection, and lead-related complications. Registry data show high procedural success and low infection rates, with serious adverse events in approximately 2-3 per cent of patients.12
Most systems provide ventricular pacing only and are therefore mainly used in patients with atrial fibrillation or infrequent pacing requirements, although newer devices can achieve AV synchrony by sensing atrial contraction via accelerometers. They are particularly useful in patients with limited venous access, prior device infection, or high infection risk, such as those on haemodialysis.
However, limited long-term randomised data and uncertainties regarding retrieval and device longevity mean they are currently implanted mainly in older patients with atrial fibrillation.13 New modular systems allow wireless pairing with subcutaneous ICDs to permit anti-tachycardia pacing, and experimental devices capable of harvesting myocardial mechanical energy for self-charging are under development.14
OVERVIEW OF ICDS
ICDs prevent sudden cardiac death by detecting and terminating malignant ventricular arrhythmias. The first human ICD implantation was performed in 1980 by Mirowski and colleagues.15 Early systems required thoracotomy with epicardial patch electrodes and abdominal pulse generators. Modern devices are now available in transvenous, subcutaneous, and extravascular configurations, although transvenous systems remain most common.16,17
These resemble pacemakers, consisting of a pectoral pulse generator connected to the right ventricle via a transvenous lead inserted through the cephalic or subclavian vein (Figure 3). On chest radiography, they may be identified by a thickened ‘shock coil’ on the right ventricular lead, which enables high-energy defibrillation. Contemporary transvenous ICD leads also provide bradycardia pacing, and many devices incorporate cardiac resynchronisation capability in combined ‘CRT-D’ systems (Figure 2).
When a malignant ventricular arrhythmia is detected, therapy is delivered after a programmed detection interval. Initial treatment often consists of anti-tachycardia pacing (ATP), which delivers rapid pacing impulses to interrupt re-entrant ventricular tachycardia. If ATP fails, or if the arrhythmia is sufficiently rapid, the device charges its capacitors and delivers a high-energy defibrillation shock.
Modern ICDs use sophisticated algorithms to distinguish ventricular arrhythmias from supraventricular rhythms and electrical noise, although detection thresholds and therapies remain programmable. Programming is therefore a critical determinant of outcomes,18 as inappropriate shocks are associated with increased morbidity and mortality.19
Approximately 20-35 per cent of patients experience at least one ICD shock during device lifetime,18 of which 10-20 per cent are inappropriate, most commonly due to atrial arrhythmias, sinus tachycardia, lead malfunction, or oversensing.19 Patients typically perceive shocks as a sudden forceful jolt lasting milliseconds.
Beyond discomfort, inappropriate shocks are associated with anxiety, reduced quality of life, depression, increased hospitalisation, and higher mortality.19,20 Repeated shocks also accelerate battery depletion, shortening generator longevity, and increasing the need for earlier replacement.
ICD indications: Approximately 70-80 per cent of ICDs are implanted for primary prevention in patients without prior ventricular arrhythmia but at high-risk of sudden cardiac death, most commonly due to left ventricular systolic dysfunction.16 The remainder are implanted for secondary prevention in survivors of sustained ventricular tachycardia, ventricular fibrillation, or aborted cardiac arrest (Table 3).
Primary prevention: Patients with symptomatic heart failure and reduced ejection fraction (typically LVEF) ≤35%) are at increased risk of sudden cardiac death, and ICD therapy reduces arrhythmic death and, in selected populations, all-cause mortality. Evidence is strongest in ischaemic cardiomyopathy, where multiple randomised controlled trials (RCTs) demonstrate mortality benefit,21,22 although implantation should be deferred until ≥40 days after myocardial infarction (MI),23 following neutral early post-MI trials.24,25
In non-ischaemic cardiomyopathy, ICD therapy consistently reduces sudden cardiac death, although effects on all-cause mortality have been less consistent,26 likely reflecting competing non-arrhythmic mortality, widespread use of CRT, and improvements in contemporary heart failure therapy. European Society of Cardiology (ESC) guidelines therefore frame recommendations more cautiously in this group.23
Regardless of aetiology, patients should receive at least three months of maximally tolerated guideline-directed medical therapy before ICD consideration, as reverse remodelling may improve LVEF and obviate the need for device therapy.23 It should be noted that most RCTs supporting ICD therapy were conducted before the widespread use of many contemporary heart failure treatments.
As such, several trials are currently underway to reassess the benefit of ICDs in the context of modern guideline-directed heart failure therapy.27 In the existing trials, survival curves typically diverge only after the first year, so candidates should have a reasonable life expectancy beyond one year.23
The same principle applies at the time of generator replacement. An ICD that was appropriately indicated earlier in a patient’s disease trajectory may confer diminishing benefit with advancing age, increasing comorbidity, or competing non-arrhythmic mortality. As generator change is not without procedural risk, it is recommended that a shared decision-making discussion take place with the patient and their next of kin regarding reassessment of ongoing indication and replacement take place, rather than automatic device renewal.
Outside heart failure populations, evidence guiding ICD implantation is more limited, particularly in inherited cardiomyopathies and channelopathies where sudden cardiac death risk may be substantial, but LVEF is less informative. In hypertrophic cardiomyopathy, ICD implantation is considered when estimated five-year sudden cardiac death risk exceeds 6 per cent, with 4-6 per cent representing intermediate-risk requiring shared decision-making.23
In inherited channelopathies such as long QT syndrome, Brugada syndrome, and catecholaminergic polymorphic ventricular tachycardia, ICD therapy is primarily indicated for secondary prevention, although selected high-risk patients with recurrent syncope or ventricular arrhythmias despite therapy may be considered. In cardiac sarcoidosis, ICD implantation is recommended for sustained ventricular arrhythmias or LVEF ≤35 per cent, and considered with high-grade AV block, unexplained syncope, or inducible ventricular tachycardia.23
In myocarditis, implantation is usually deferred during the acute phase. Myocardial scar on cardiac MRI may further refine risk stratification.23,28
Secondary prevention: ESC guidelines recommend ICD implantation for secondary prevention in survivors of ventricular fibrillation or haemodynamically significant sustained ventricular tachycardia when no reversible cause is identified and life expectancy exceeds one year. Reversible triggers, including acute myocardial ischaemia, electrolyte disturbance, drug toxicity, or acute myocarditis, should first be corrected, with reassessment of arrhythmic risk once the acute condition has resolved.23
Complications of CIED implantation
Complications may occur during implantation or accumulate over the lifetime of the device. Major complications of transvenous CIED implantation occur in approximately 3-8 per cent of cases, depending on device type and population.29 These include access-related complications such as pneumothorax or haemothorax, pocket haematoma, and lead-related problems including dislodgement or failure of capture.
Less commonly, myocardial perforation with pericardial effusion or tamponade may occur. Women have a 30-50 per cent higher risk of device-related complications, likely reflecting smaller vascular and cardiac anatomy, thinner myocardial walls, and lower body mass.30 Early device infection may also occur, although less common. Most complications are manageable when recognised promptly.31
Risk does not end after implantation and increases over time due to lead failure, infection, and generator replacement procedures.30,32 Device infection is particularly serious and occurs most commonly following generator changes or secondary to bacteraemia.
Confirmed infection generally requires complete system explant, as antibiotics alone rarely achieve eradication.3 Extraction of long-standing transvenous systems can carry a small, but real, mortality risk because leads become fibrotically integrated within venous and cardiac structures.33 This is particularly relevant in younger patients who may require multiple generator replacements during their lifetime.
Patients receiving haemodialysis require special consideration, as CIED implantation carries higher risks of infection and vascular access complications. Preservation of central venous access is therefore important.
Routine primary prevention ICD implantation in dialysis patients has not consistently demonstrated survival benefit, and decisions should be individualised, although secondary-prevention indications remain stronger.34
SUBCUTANEOUS ICD (S-ICD)
To avoid complications related to intravascular leads, the S-ICD was introduced in 2010. The generator is implanted in a lateral thoracic pocket and connected to a subcutaneous lead tunnelled parallel to the sternum containing the defibrillation coil.
Clinical trials and registry data demonstrate similar efficacy to transvenous ICDs for terminating ventricular arrhythmias, although higher energy requirements make the device larger. Unlike transvenous systems, S-ICDs cannot provide chronic bradycardia pacing or anti-tachycardia pacing.
Emerging strategies combine S-ICDs with leadless pacemakers, allowing limited pacing capability while maintaining a completely extravascular defibrillation system.
EXTRAVASCULAR ICD
More recently, extravascular ICD systems have been developed to combine the advantages of entirely extravascular defibrillation with the ability to deliver ATP. In these systems, the generator is implanted in the left lateral thoracic region, similar to a S-ICD, while a single lead is positioned within the substernal space, anterior to the heart, but outside the vasculature. This location allows more effective sensing and pacing than purely subcutaneous systems while avoiding the long-term complications associated with transvenous leads.
Early clinical studies have demonstrated effective defibrillation and the ability to deliver anti-tachycardia pacing for ventricular tachycardia, with high procedural success rates.35 Although longer-term outcome data are still emerging, extravascular ICDs may represent an important intermediate strategy between transvenous and subcutaneous systems, particularly in younger patients or those at higher risk of lead-related complications. Concerns remain around how these leads could be extracted if needed.
IMPLANTED CARDIAC MONITORING DEVICES (ICMDS)
ICMDs are small devices (6-7mm; approximate weight of 2g) inserted subcutaneously in the left pectoral region that provide continuous rhythm monitoring for the lifetime of the device, typically two to three years.¹⁹ Using the same remote monitoring infrastructure as ICDs and some pacemakers, they enable detection of infrequent, but clinically important, arrhythmias such as sinus pauses, intermittent AV block, atrial fibrillation, and ventricular arrhythmias.
Implantable loop recorders are most commonly used in the evaluation of recurrent unexplained syncope when a cardiac cause is suspected, where prolonged monitoring substantially increases diagnostic yield.19
However, they are relatively expensive and, unlike other implantable rhythm devices, cannot deliver therapy, which may be a limitation when malignant ventricular arrhythmia is strongly suspected.
For patients with intermittent, but symptomatic palpitations, wearable ECG-capable devices, such as smartwatch-based monitors, may be more appropriate as they allow patient-triggered recordings during symptoms and facilitate symptom-rhythm correlation without an invasive implant.
REMOTE MONITORING
Most modern ICDs, implantable cardiac monitors, and many pacemakers support remote monitoring using a home transmitter, typically kept beside the patient’s bed. These systems communicate automatically with the device at scheduled intervals or after clinically important events, such as ICD therapy or detection of significant arrhythmia.
Remote monitoring allows clinicians to assess device function and arrhythmia episodes without routine in-person visits and has been shown to facilitate earlier detection of device malfunction, lead failure, and clinically significant arrhythmias, while reducing hospital attendance.3
MRI COMPATIBILITY
As both CIED implantation and MRI utilisation increase,36,37 device safety during MRI has become increasingly important. Historically, MRI was avoided due to concerns regarding lead heating, device movement, and electromagnetic interference, which could lead to device malfunction, reset of programming settings, inappropriate pacing inhibition, or shocks.37
Modern systems are therefore designed to be MRI-conditional, meaning scanning can be performed safely under specific conditions including device interrogation and reprogramming, temporary suspension of ICD therapies, physiological monitoring during scanning, and post-scan device reassessment. The term ‘MRI-conditional’ is preferred, rather than ‘MRI-compatible’ or ‘MRI-safe’, emphasising that safety depends on adherence to structured protocols, rather than just the physical properties of the device.
CIEDs also contain magnetic sensors that respond to strong external magnetic fields. In pacemakers, magnet application typically results in asynchronous pacing, preventing inappropriate inhibition, while in ICDs it suspends tachyarrhythmia detection and shock therapy without altering pacing. This feature is commonly used as an emergency measure to temporarily prevent inappropriate or repetitive ICD shocks.
Increasing evidence suggests however that even non-MRI-conditional systems, including legacy devices or mixed-manufacturer systems,37,38,39 can often be scanned safely when clinically necessary and under appropriate protocols.23 However, abandoned, damaged, or epicardial leads may be more prone to heating and require greater caution.39
| CLINICAL SETTING | ICD RECOMMENDED | ICD SHOULD BE CONSIDERED |
ICD NOT RECOMMENDED |
|---|---|---|---|
| Secondary prevention | Survivors of cardiac arrest due to VT/VF without reversible cause |
Arrhythmia due to reversible causes (eg, acute ischaemia, electrolyte disturbance, drug toxicity) |
|
| Ischaemic cardiomyopathy | LVEF ≤35% despite ≥3 months optimal medical therapy |
LVEF ≤40% with NSVT and inducible VT on electrophysiology study |
Early after MI (<40 days) |
| Non-ischaemic cardiomyopathy |
LVEF ≤35% despite optimal medical therapy |
LVEF <50% with additional high-risk features (eg, syncope, LGE on CMR, pathogenic mutations) |
Transient cardiomyopathy expected to recover |
| Inherited cardiomyopathies (HCM, ARVC) |
High estimated sudden death risk or sustained VT |
Low-risk phenotype without risk markers |
|
| Channelopathies (LQTS, Brugada, CPVT) |
Survivors of cardiac arrest | High-risk patients despite optimal medical therapy |
Asymptomatic low-risk genotype |
| General considerations | Life expectancy <1 year or severe competing comorbidity |
TABLE 3: Indications for ICD implantation, stratified by clinical scenario, and recommendation strength.
ICD = Implantable cardiac defibrillator; CMR = Cardiac magnetic resonance imaging; LVEF = Left ventricular ejection fraction; VT = Ventricular tachycardia; VF = Ventricular fibrillation; HCM = Hypertrophic cardiomyopathy; ARVC = Arrhythmogenic right ventricular cardiomyopathy; LGE = Late gadolinium enhancement; LQTS = Long QT syndrome; CPVT = Catecholaminergic polymorphic ventricular tachycardia.
Adapted from: 2022 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death
PERI-OPERATIVE MANAGEMENT OF ICDS
Electromagnetic interference during surgery, most commonly from electrocautery, can theoretically inhibit pacemaker output or trigger inappropriate ICD therapies. The risk is low with bipolar cautery or procedures below the umbilicus.40 When unipolar cautery is required, the dispersive pad should be positioned as far from the device as possible.
Current guidelines recommend pre-operative device assessment, temporary asynchronous pacing in pacing-dependent patients, suspension of ICD therapies during procedures with significant electromagnetic interference, continuous monitoring, and post-operative device reassessment.40
Conclusion
CIEDs have become integral to contemporary cardiovascular care, providing effective therapy for bradyarrhythmias, prevention of sudden cardiac death, and treatment of selected heart failure populations. As implantation rates continue to rise and device technologies evolve, familiarity with device indications, complications, and practical management considerations is increasingly important for clinicians across a wide range of specialties.
References
- Schechter DC. Background of clinical electrostimulation. VII. Modern era of artificial cardiac pacemakers. N Y State J Med. 1972;72(10):1166-1191.
- Kelly R. Heart Rhythm International – AI enabled cardiac rhythm management system. Available at: https://heartrhythmireland.com/.
- Glikson M, Nielsen JC, Kronborg MB, et al. 2021 ESC Guidelines on cardiac pacing and cardiac resynchronisation therapy. Europace. 2022;24(1):71-164. doi:10.1093/europace/euab232.
- Brubaker PH, Kitzman DW. Chronotropic incompetence: Causes, consequences, and management. Circulation. 2011;123(9):1010-1020. doi:10.1161/CIRCULATIONAHA.110.940577.
- Kuroda S, Kawamura I, Sahashi Y, et al. Effectiveness of rate-adaptive pacing on patients with chronotropic incompetence: Systematic review and meta-analysis of randomised controlled trials. Int J Cardiol. 2025;423:133022. doi:10.1016/j.ijcard.2025.133022.
- Reddy YNV, Koepp KE, Carter R, et al. Rate-adaptive atrial pacing for heart failure with preserved ejection fraction: The RAPID-HF randomised clinical trial. JAMA. 2023;329(10):801-809. doi:10.1001/jama.2023.0675.
- Shah Syed AR, Akram A, Azam MS, et al. Dual-chamber versus single chamber pacemakers, a systemic review and meta-analysis on sick sinus syndrome and atrioventricular block patients. Heliyon. 2023;10(1):e23877. doi:10.1016/j.heliyon.2023.e23877.
- Sweeney MO, Prinzen FW. A new paradigm for physiologic ventricular pacing. J Am Coll Cardiol. 2006;47(2):282-288. doi:10.1016/j.jacc.2005.09.029.
- Cleland JG, Daubert JC, Erdmann E, et al. The effect of cardiac resynchronisation on morbidity and mortality in heart failure. N Engl J Med. 2005;352(15):1539-1549. doi:10.1056/NEJMoa050496.
- Young JB, Abraham WT, Smith AL, et al. Combined cardiac resynchronization and implantable cardioversion defibrillation in advanced chronic heart failure: The MIRACLE ICD Trial. JAMA. 2003;289(20):2685-2694. doi:10.1001/jama.289.20.2685.
- Bristow MR, Saxon LA, Boehmer J, et al. Cardiac-resynchronisation therapy with or without an implantable defibrillator in advanced chronic heart failure. N Engl J Med. 2004;350(21):2140-2150. doi:10.1056/NEJMoa032423.
- El-Chami MF, Garweg C, Clementy N, et al. Leadless pacemakers at five-year follow-up: The Micra transcatheter pacing system post-approval registry. Eur Heart J. 2024;45(14):1241-1251. doi:10.1093/eurheartj/ehae101.
- Roberts PR, Clémenty N, Mondoly P, et al. A leadless pacemaker in the real-world setting: Patient profile and performance over time. J Arrhythm. 2023;39(1):1-9. doi:10.1002/joa3.12811.
- Liu Z, Hu Y, Qu X, et al. A self-powered intracardiac pacemaker in swine model. Nat Commun. 2024;15(1):507. doi:10.1038/s41467-023-44510-6.
- Ammannaya GKK. Implantable cardioverter defibrillators – the past, present and future. Arch Med Sci Atheroscler Dis. 2020;5:e163-e170. doi:10.5114/amsad.2020.97103.
- Yousuf OK, Kennedy K, Russo A, et al. Appropriateness of implantable cardioverter-defibrillator device implants in the US. Heart Rhythm. 2024;21(4):397-407. doi:10.1016/j.hrthm.2023.12.005.
- Knops RE, Olde Nordkamp LRA, Delnoy PHM, et al. Subcutaneous or transvenous defibrillator therapy. N Engl J Med. 2020;383(6):526-536. doi:10.1056/NEJMoa1915932.
- Moss AJ, Schuger C, Beck CA, et al. Reduction in inappropriate therapy and mortality through ICD programming. N Engl J Med. 2012;367(24):2275-2283. doi:10.1056/NEJMoa1211107.
- Daubert JP, Zareba W, Cannom DS, et al. Inappropriate implantable cardioverter-defibrillator shocks in MADIT II: Frequency, mechanisms, predictors, and survival impact. J Am Coll Cardiol. 2008;51(14):1357-1365. doi:10.1016/j.jacc.2007.09.073.
- Poole JE, Johnson GW, Hellkamp AS, et al. Prognostic importance of defibrillator shocks in patients with heart failure. N Engl J Med. 2008;359(10):1009-1017. doi:10.1056/NEJMoa071098.
- Moss AJ, Zareba W, Hall WJ, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med. 2002;346(12):877-883. doi:10.1056/NEJMoa013474.
- Bardy GH, Lee KL, Mark DB, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med. 2005;352(3):225-237. doi:10.1056/NEJMoa043399.
- Zeppenfeld K, Tfelt-Hansen J, de Riva M, et al. 2022 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur Heart J. 2022;43(40):3997-4126. doi:10.1093/eurheartj/ehac262.
- Hohnloser SH, Kuck KH, Dorian P, et al. Prophylactic use of an implantable cardioverter-defibrillator after acute myocardial infarction. N Engl J Med. 2004;351(24):2481-2488. doi:10.1056/NEJMoa041489.
- Steinbeck G, Andresen D, Seidl K, et al. Defibrillator implantation early after myocardial infarction. N Engl J Med. 2009;361(15):1427-1436. doi:10.1056/NEJMoa0901889.
- Køber L, Thune JJ, Nielsen JC, et al. Defibrillator implantation in patients with nonischemic systolic heart failure. N Engl J Med. 2016;375(13):1221-1230. doi:10.1056/NEJMoa1608029.
- Dagres N, Merkely B, Hindricks G. An update on the PROFID Project 2025. Eur Heart J. 2026;47(6):660-661. doi:10.1093/eurheartj/ehaf845.
- Śpiewak M, Kłopotowski M, Ojrzyńska N, et al. Impact of cardiac magnetic resonance on the diagnosis of hypertrophic cardiomyopathy – a 10-year experience with over 1,000 patients. Eur Radiol. 2021;31(3):1194-1205. doi:10.1007/s00330-020-07207-8.
- Poole JE, Gleva MJ, Mela T, et al. Complication rates associated with pacemaker or implantable cardioverter-defibrillator generator replacements and upgrade procedures: Results from the REPLACE registry. Circulation. 2010;122(16):1553-1561. doi:10.1161/CIRCULATIONAHA.110.976076.
- Uslan DZ, Gleva MJ, Warren DK, et al. Cardiovascular implantable electronic device replacement infections and prevention: Results from the REPLACE Registry. Pacing Clin Electrophysiol. 2012;35(1):81-87. doi:10.1111/j.1540-8159.2011.03257.x.
- Burri H, Starck C, Auricchio A, et al. EHRA expert consensus statement and practical guide on optimal implantation technique for conventional pacemakers and implantable cardioverter-defibrillators: Endorsed by the Heart Rhythm Society (HRS), the Asia Pacific Heart Rhythm Society (APHRS), and the Latin-American Heart Rhythm Society (LAHRS). Europace. 2021;23(7):983-1008. doi:10.1093/europace/euaa367.
- Kirkfeldt RE, Johansen JB, Nohr EA, Nohr EA, et al. Complications after cardiac implantable electronic device implantations: An analysis of a complete, nationwide cohort in Denmark. Eur Heart J. 2014;35(18):1186-1194. doi:10.1093/eurheartj/eht511.
- Kusumoto FM, Schoenfeld MH, Wilkoff BL, et al. 2017 HRS expert consensus statement on cardiovascular implantable electronic device lead management and extraction. Heart Rhythm. 2017;14(12):e503-e551. doi:10.1016/j.hrthm.2017.09.001.
- Wright DJ, Trucco ME, Zhou J, et al. Chronic kidney disease and transvenous cardiac implantable electronic device infection – is there an impact on healthcare utilisation, costs, disease progression, and mortality? Europace. 2024;26(7):euae169. doi:10.1093/europace/euae169.
- Friedman P, Murgatroyd F, Boersma LVA, et al. Performance and safety of the extravascular implantable cardioverter defibrillator through long-term follow-up: Final results from the pivotal study. Circulation. 2025;151(4):322-332. doi:10.1161/CIRCULATIONAHA.124.071795.
- OECD. Diagnostic technologies: Health at a glance 2023. Available at: www.oecd.org/en/publications/health-at-a-glance-2023_7a7afb35-en/full-report/diagnostic-technologies_23088ad0.html.
- Lanz H, Strauß K, Höpler J, et al. Safety of magnetic resonance imaging in patients with cardiac implantable electronic devices. J Cardiovasc Dev Dis. 2024;11(10):313. doi:10.3390/jcdd11100313.
- Russo RJ, Costa HS, Silva PD, et al. Assessing the risks associated with MRI in patients with a pacemaker or defibrillator. N Engl J Med. 2017;376(8):755-764. doi:10.1056/NEJMoa1603265.
- Bhuva AN, Moralee R, Brunker T, et al. Evidence to support magnetic resonance conditional labelling of all pacemaker and defibrillator leads in patients with cardiac implantable electronic devices. Eur Heart J. 2022;43(26):2469-2478. doi:10.1093/eurheartj/ehab350.
- Thomas H, Plummer C, Wright IJ, Foley P, Turley AJ. Guidelines for the peri-operative management of people with cardiac implantable electronic devices: Guidelines from the British Heart Rhythm Society. Anaesthesia. 2022;77(7):808-817. doi:10.1111/anae.15728.