The incidence and mortality rate of metastatic melanoma continue to rise but treatment is improving, write Dr Lisa Roche, Dr Catherine Foley and Dr Michelle Murphy
Case report 1
A 30-year-old woman with multiple moles was referred to dermatology with a changing mole on her back. She was concerned that a long-standing mole on her right upper back had increased in size, become darker in colour and developed black and red areas at the edges over the preceding four months. The patient had a history of blistering sunburn in childhood, sunbed use in adulthood, and fair skin. There was no family history of melanoma and she had never been immunosuppressed.
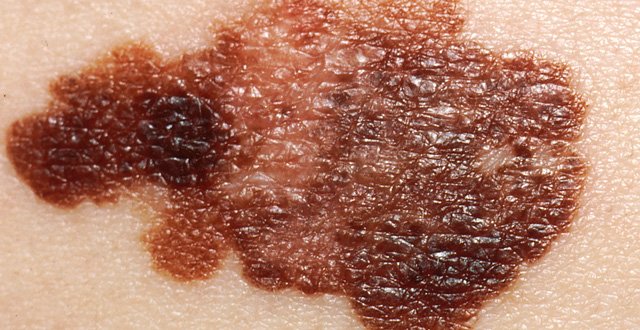
The lesion on the right upper back measured 1cm in diameter, was asymmetrical with irregular borders, and was multi-coloured, with brown, red and black areas (Figure 1). An excision biopsy was performed on the same day and the lesion was excised, with a 2mm clinical margin and a cuff of subcutaneous fat.
Histopathological evaluation showed a malignant melanoma, superficial spreading type, with a Breslow depth of 1.25mm and a mitotic count of 2/mm2. There was no ulceration of overlying epidermis.
Following discussion at the regional skin cancer multidisciplinary team (MDT) meeting, the patient was referred to plastic surgery for a wide local excision with 2cm margins and a sentinel node biopsy. There was no residual melanoma present in the wide local excision specimen and two sentinel nodes were identified but did not contain evidence of metastases on histopathological or immunohistochemistry evaluation.
The patient was given verbal and written education on melanoma, changes to look for in moles (ABCDE criteria), technique for regular self-skin examination, photoprotection and vitamin D supplementation. Full skin examination and assessment for lymphadenopathy and organomegaly is performed at three-monthly intervals until three years have passed from the date of diagnosis.
Case report 2
A 74-year-old man presented to his GP with a lesion on his lower back. Initially, the lesion was thought to be a seborrheic keratosis and had been treated with cryotherapy. On review, there was concern about pigmentation at the base of the lesion and the patient was referred to the regional pigmented lesion clinic.
There was no history of occupational sun exposure, no family history of melanoma, no sunbed use and the patient went on two sun holidays a year. Clinically, the lesion measured 5mm and had pale and dark areas (Figure 2).
There was a blue veil seen on dermoscopy.
The lesion was excised with a 2mm clinical margin and histopathology showed a melanoma, Breslow depth 0.8mm, with no adverse prognostic features (ie, ulceration or mitoses).
The patient had a subsequent wide local excision with a 1cm margin four weeks later and there was no residual melanoma seen. His serum vitamin D level was reduced at 34nmol/L (normal range >50nmol/L). The patient was reviewed in dermatology outpatients every three months for 12 months following diagnosis. During this time, he was educated on self-skin checks, changes to look for in moles and the importance of photoprotection. Full skin examination and clinical evaluation of regional lymph node basins were performed at every visit.
The incidence of malignant melanoma is increasing worldwide, particularly in countries with skin type and climate similar to Ireland. Every year, there are over 700 new cases of melanoma diagnosed. Figures from the National Cancer Registry Ireland (NCRI) show that melanoma is the fifth-most common cancer diagnosed, accounting for 4.4 per cent of all malignant neoplasms. During 1995-2007, the number of new cases diagnosed increased at approximately 5 per cent per annum overall. Mortality from melanoma is increasing and currently, approximately 160 deaths occur from melanoma each year. This is an increase from under 60 deaths per year in the mid-1990s and from less than 20 deaths per year during the 1950s.
Symptoms
When evaluating a mole for melanoma, the ABCDE criteria are significant. The ABCD mnemonic (asymmetry, border, colour variegation, diameter >6mm) was published in 1985 in an effort to educate doctors and the public on changes that might indicate melanoma in early clinical stages. It has been a very successful tool and the depth of melanoma diagnosed in the US is now much thinner than prior to its introduction. Not all melanomas will be picked up by ABCD and therefore, history of evolution can be an important clue. The letter ‘E’, for evolution, was added in 2004 when it was recognised that changes in appearance or symptomatology of moles may indicate melanoma. Evolution can be an important signal, especially for nodular, desmoplastic and amelanotic melanomas, which may not be picked up using just the ABCD mnemonic and which often present as thick, dangerous lesions. For patients to be aware of changes in pre-existing naevi, it is important to encourage regular self-skin checks. The ‘ugly duckling’ sign, ie, a naevus out of keeping with an individual’s mole pattern, is another useful tool in assessing naevi.
Diagnosis
GPs are the first point of medical contact for the majority of patients with suspicious skin lesions. The National Cancer Control Programme (NCCP) produced pigmented lesion referral guidelines in 2010. A national referral form has been launched recently to facilitate rapid access to consultant dermatologists and plastic surgeons for patients with lesions clinically suspicious for melanoma based on known patient risk factors (Table 1) and suspicious features of individual lesions.
The processes underlying the development of melanoma are complex and vary between individuals. NCCP guidelines suggest that patients with suspicious pigmented lesions should be referred to dermatology or plastic surgery with the lesion intact. Pigmented lesion clinics have been set up in many centres to facilitate rapid access for these patients. Best practice is that skin lesions suspected of being melanoma should be excised completely with a 2-3mm clinical margin, and a cuff of fat. This allows for accurate measurement of the Breslow depth and planning of definitive treatment. All specimens should be sent for histological analysis. All invasive melanomas should be discussed at the regional melanoma MDT meeting.
Treatment
Further treatment is decided based on the histopathological features of the tumour after discussion at the regional skin cancer MDT meeting. For all patients with in situ or invasive disease, a wide local excision (WLE) of the scar is performed, the extent of which depends on the Breslow depth of the tumour. For patients with thin melanoma, ie, <1mm, this may be curative and is the only treatment required. Sentinel lymph node biopsy is usually offered to patients with melanoma of ≥1mm depth. This procedure involves injection of a radiolabelled dye to the scar from the primary excision and subsequent identification of the first node that area drains to in the regional lymph node basin. Following WLE, all cases are further discussed at the regional skin cancer MDT meeting and follow-up is recommended based on this.
Sentinel node biopsy offers prognostic information, but its role in improving outcomes is controversial. There is no role for staging investigations in early disease, but they may be performed on the advice of the MDT meeting for more advanced disease.
The treatment of metastatic melanoma has undergone a revolution in the last 10 years with the development of checkpoint inhibition and targeted agents that may improve survival from metastatic disease. As melanoma has been found to be one of the most sensitive malignancies to immune modulation, the development of immunotherapies such as ipilimumab, anti-CTLA4, and newer anti-programmed death 1 agents (nivolumab, pembrolizumab) have changed the outlook for patients with metastatic melanoma and offer hope of prolonged overall survival. Up to 50 per cent of cutaneous melanomas carry a mutation in the BRAF gene, and in this group, ipilimumab in combination with anti-PD-1 based therapies demonstrates further improvement in overall survival. The anti-PD-1-based therapies (nivolumab, pembrolizumab) can also be used as single agents in wild-type (non-mutated) melanoma or in cases when BRAF status is unknown, to achieve improved survival. A conservative estimate for expected five-year survival for metastatic melanoma is currently around 35 per cent however, this could be as high as 50 per cent in the combination therapy groups (nivolumab/ipilimumab).
For metastatic melanoma that is known to have a specific BRAF V600 driver mutation, treatment achieved by targeting the MAP (mitogen-activated protein) kinase pathway with a combination of a BRAF inhibitor and a MEK inhibitor is another treatment option. Based upon large phase 3 trials, a combination of dabrafenib (a BRAF inhibitor) and trametinib (a MEK inhibitor) is approved for adjuvant use in melanoma with lymph node involvement and a BRAF V600E or V600K mutation.
Not unsurprisingly, checkpoint inhibition is associated with a wide spectrum of side-effects, in this case termed ‘immune-related adverse events’. These are thought to arise secondary to enhancement immune activation and have been known to affect the skin, gastrointestinal tract, hepatic and endocrine systems.
Short-term immunosuppression, often with corticosteroids, is first-line treatment, however rare and even fatal toxicities have been reported.
Current/future trials
Many large stage 3 trials are ongoing, examining combinations of the above treatments, as well as establishing the most beneficial sequencing and duration of therapy of these agents in advanced melanoma. Novel agents are also in the pipeline. The overall aim and current challenge in this field is gaining an improved understanding of the immune-biology of melanoma; facilitating the development of individualised treatment regimens for patients, tailored to maximise clinical benefit and decrease toxicity. This will hopefully be achieved by optimally integrating immune and non-immune therapies, developing validated predictive biomarkers, both for metastases and in the adjuvant setting, to improve prognostics.
While our understanding of, and the treatment options for, metastatic melanoma are evolving and newer treatments offer hope of prolonged overall survival for patients, early detection still gives patients the best chance of survival.












Leave a Reply
You must be logged in to post a comment.