An overview of diagnosing and treating OAB, particularly in post-menopausal women
Overactive bladder (OAB) is a condition that can significantly impede one’s quality-of-life. While it affects a wide spectrum of individuals, post-menopausal women are particularly susceptible. This article aims to delve deeper into OAB, differentiating it from other urinary conditions, exploring its diagnostic tools, and outlining the appropriate treatment options. Special attention has been given to the post-menopausal age group to highlight their unique challenges and considerations.
Definition of OAB
OAB is a clinical syndrome characterised by symptoms like urinary urgency, often accompanied by frequency and nocturia, with or without urge urinary incontinence. It differs from stress urinary incontinence and requires a distinct approach.
Epidemiology
In Ireland alone, up to a third of the population suffers from urinary incontinence, and OAB constitutes a significant portion of these cases. Among the affected, post-menopausal women stand out as a notably at-risk subgroup. Studies indicate that among post-menopausal women, almost half report OAB symptoms, with many experiencing a significant decrease in quality-of-life. Notably, these symptoms often go unreported, attributed to age-related changes, or are simply endured in silence due to stigma or lack of awareness.
Case presentation
Ms Johnson, a 64-year-old woman, presented with a six-month history of frequent, urgent urination often leading to involuntary leakage. She described waking up three-to-four times nightly due to this urgency. She had been menopausal for 10 years and was not on HRT. During her physical examination, significant vaginal dryness was noted. Ms Johnson also reported painful intercourse. No history of UTIs was recorded, but she mentioned an increasing coffee consumption. She expressed distress over her reduced social activities, fearing sudden episodes of incontinence.
Pathophysiology
Mechanisms leading to OAB symptoms: Several aetiological factors have been postulated for OAB:
Genitourinary syndrome of menopause: The transition into menopause, marked by a decline in oestrogen levels, has a profound impact on the urogenital system. The thinning of the urethral lining, reduction in pelvic muscle tone, and decreased bladder elasticity, all often lead to increased urgency, frequency, and even incontinence.
Neurological aberrations: Neural misfires or dysfunctions can disrupt the delicate balance of storage and voiding.
Systemic illnesses: Conditions like diabetes mellitus, neurological diseases, and obesity can indirectly exert pressure on bladder function.
Medicinal impact: Some medications have diuretic properties or can interfere with nerve signaling, leading to incontinence.
Structural abnormalities: Prolapse or any distortions in the bladder or its outflow system can precipitate OAB.
Cognitive decline: Age-associated cognitive deficiencies can lead to an impaired sense of bladder fullness or voiding needs.
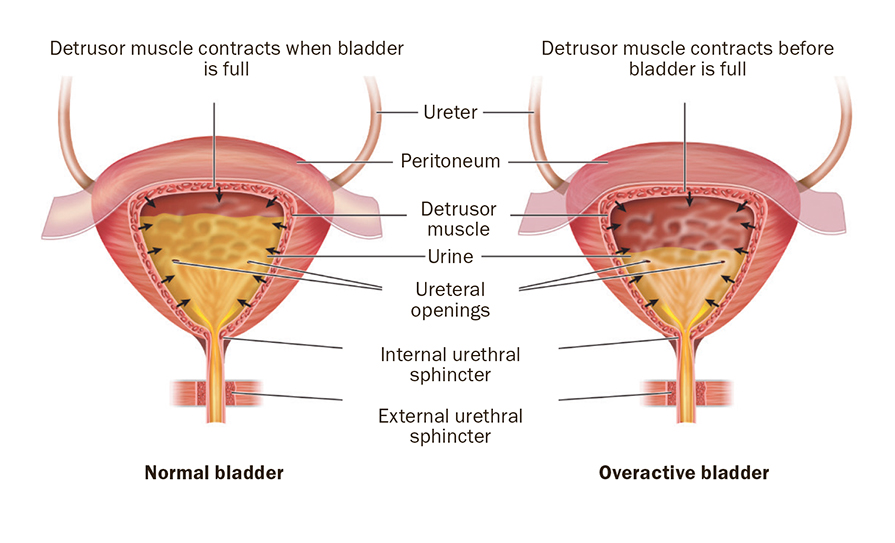
Figure 1: Normal bladder versus overactive bladder
Clinical presentation
Key symptoms: Urgency, frequency, nocturia, with or without urge incontinence
While OAB predominantly manifests as a sudden, intense need to urinate, the clinical presentation can be varied. This intense urge is frequently accompanied by an increase in the frequency of voiding, both during the day and night. Nocturia specifically can disrupt sleep patterns, leading to chronic fatigue and its consequent challenges. Urge incontinence, or the sudden, uncontrollable leakage of urine following an intense urge to void, is among the most distressing symptoms.
Differentiating between OAB and other urinary conditions: Urinary incontinence is an umbrella term with various subtypes:
Stress urinary incontinence (SUI): This arises when external pressure on the bladder, such as laughing or coughing, leads to leakage.
Urge urinary incontinence (UUI): Characterised by a sudden urge followed by involuntary urination.
Mixed incontinence: A combination of SUI and urge incontinence, often reflecting a multifactorial aetiology.
Diagnostic tools
Patient history: A detailed history lays the foundation. This encompasses:
Symptom onset and progression: Determining whether the onset was sudden or gradual, identifying potential triggers, gauging the frequency of episodes both during the day and night, noting any associated symptoms or factors, such as recurrent UTIs and haematuria. Additionally, understanding any relation between sexual function and incontinence, particularly in post-menopausal women, can offer valuable insights.
Other medical conditions: Any history of neurological disorders or diabetes.
Medication review: Are they on diuretics, antihistamines, or any other medication that might precipitate or exacerbate incontinence.
Bladder diary: A bladder diary serves as an invaluable window into the patient’s daily life, and includes:
Fluid intake patterns: Consumption of bladder irritants like caffeine.
Voiding schedules: Pattern to urgency/incontinence episode.
Incontinence episodes: Can hint towards potential triggers.
Physical examination: This involves:
Abdominal and pelvic exams: To rule-out masses, tenderness, prolapse or any structural abnormalities.
Neurological examination: Ensuring the neural pathways governing bladder control are intact.
Urine analysis: This simple test can unearth infections, blood (haematuria), or other abnormalities that might be masquerading as or exacerbating OAB.
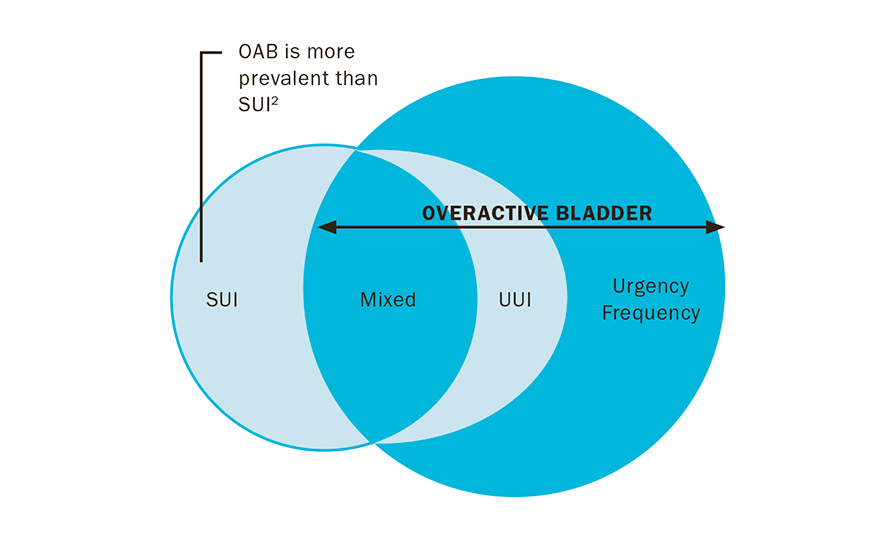
Figure 2: OAB and urinary incontinence
Treatment approaches
Lifestyle and behavioural interventions:
Dietary modifications: Substances like caffeine, found in coffee, tea, and many soft drinks, as well as alcohol, are known irritants for the bladder. Their diuretic properties can exacerbate OAB symptoms. Reducing or eliminating these from the diet can significantly ameliorate the symptoms, allowing patients greater comfort and fewer unscheduled bathroom visits.
Timed voiding and bladder training: One of the keys to managing OAB effectively is to retrain the bladder. This involves setting a urination schedule, progressively increasing the time between each visit. Over time, this can extend the bladder’s capacity and reduce the urgency and frequency of urination, granting patients a greater degree of autonomy over their bladder functions.
Pelvic floor exercises: The pelvic floor muscles are a critical support system for the bladder. Strengthening these muscles can significantly enhance bladder control. Engaging in exercises, often referred to as Kegel exercises, can fortify these muscles. Many patients benefit from consulting with specially trained physiotherapists who can provide guidance on correctly performing these exercises, ensuring maximum efficacy.
Sleep hygiene: Addressing nocturia often requires enhancing sleep routines. Consistent sleep schedules, a tranquil bedroom environment, limiting evening fluids, reducing screen exposure, and adopting relaxation techniques before bed can all mitigate sleep disturbances that exacerbate nocturnal bladder symptoms.
Pharmacotherapy: Medications can offer relief, but it is essential to choose wisely and be cognisant of potential side-effects.
Antimuscarinic drugs. These agents block muscarinic receptor stimulation by acetylcholine and reduce smooth muscle contraction of the bladder. Such blockade during bladder storage results in increased bladder capacity and decreased urgency. Examples of drugs in that big group family are: Fesoterodine, solifenacin, and oxybutynin patches. Common adverse effects include dry mouth, dry eye, and constipation.
Beta-3 adrenergic agonist drugs. Mirabegron works by stimulating the receptors in the bladder responsible for smooth muscle relaxation. The main advantage of this drug is the reduced side-effects, which increases tolerance for this treatment.
Vaginal oestrogen. For post-menopausal women, vaginal dryness and urinary symptoms are often interconnected, complicating OAB management. Vaginal estrogen, administered directly to the vaginal tissues, focuses on rejuvenating the vaginal and urinary tissues by restoring elasticity and lubrication. This not only alleviates vaginal dryness and discomfort, but can also improve OAB symptoms by rejuvenating the urinary tract tissues. Topical oestrogens are available in various forms, including creams, pessaries, or rings. They are generally well-tolerated, and the localised delivery minimises systemic absorption, reducing potential risks associated with systemic hormone replacement therapy (HRT).
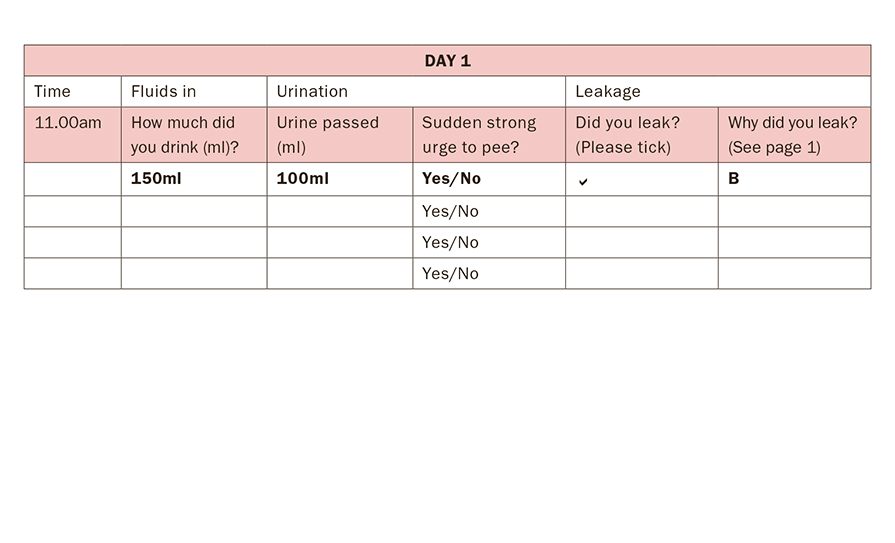
Figure 3: Bladder diary
Referral criteria
While primary care practitioners can manage many cases of OAB, certain situations warrant specialist intervention. This includes individuals who do not respond to first-line treatments, those presenting with blood in the urine (haematuria), those with significant residual urine post-voiding, or those with recurrent urinary tract infections. Additionally, significant pelvic organ prolapse, bladder pain, or other complicating factors might necessitate a referral to a specialist in urology or urogynaecology.
Second-line treatments
Posterior tibial nerve stimulation (PTNS): A minimally-invasive treatment that involves the insertion of a small, thin needle near the ankle. A device sends specific electrical impulses to the nerve pathways that control the bladder and pelvic floor. Sessions typically last about 30 minutes and are conducted weekly for an initial period.
Botox (onabotulinumtoxinA): This therapeutic modality involves injecting onabotulinumtoxinA directly into the bladder muscle, helping to relax it and thus reducing the symptoms of urgency. There are potential side-effects, including urinary retention that requires self-catheterisation.
Sacral nerve stimulation (SNS): A small device is surgically placed under the skin and acts as a ‘bladder pacemaker’. This device sends electrical pulses to the sacral nerves, helping modulate the signals that cause OAB. It is particularly beneficial for those who have not found relief from other treatment modalities.
Conclusion
OAB can have profound implications on one’s quality-of-life. As such, a nuanced understanding of its presentation, diagnostics, and treatment is critical for effective management. Both primary care providers and specialists play a pivotal role in its holistic treatment. Through a combination of medical intervention and lifestyle modifications, individuals suffering from OAB can hope for a significant improvement in their daily lives.
DR REUT ROTEM, Subspecialty Fellow in Urogynaecology, Urogynaecology Department, Cork University Maternity Hospital (CUMH); DR SUMAIYA AL SHUKAILI, Subspecialty Fellow in Urogynaecology, Urogynaecology Department, CUMH; DR SIMON CRAVEN, SpR in Obstetrics and Gynaecology, CUMH; and DR SUZANNE O’SULLIVAN, Consultant Obstetrician and Gynaecologist, CUMH












Leave a Reply
You must be logged in to post a comment.